A Modern FBA Protocol for Analyzing the E. coli Phosphotransferase System (PTS): From Core Metabolism to Advanced Modeling
This article provides a comprehensive protocol for applying Flux Balance Analysis (FBA) to study the phosphotransferase system (PTS) in Escherichia coli.
A Modern FBA Protocol for Analyzing the E. coli Phosphotransferase System (PTS): From Core Metabolism to Advanced Modeling
Abstract
This article provides a comprehensive protocol for applying Flux Balance Analysis (FBA) to study the phosphotransferase system (PTS) in Escherichia coli. Tailored for researchers and scientists, the guide covers foundational PTS biology, step-by-step FBA methodology, strategies for model troubleshooting and optimization, and advanced techniques for validating and comparing predictions with experimental data. By integrating the latest structural insights and computational approaches, this resource aims to equip professionals with the tools to accurately model PTS-mediated transport and its profound impact on carbon metabolism, regulatory networks, and biotechnological applications.
Understanding the E. coli PTS: Structure, Function, and Metabolic Role
The bacterial Phosphotransferase System (PTS) is a remarkable multi-functional complex that serves as the primary transport system for numerous carbohydrates in bacteria such as Escherichia coli [1]. This system represents a sophisticated biological solution to the challenge of nutrient acquisition, performing group translocation where sugars are phosphorylated concurrently with their translocation across the cytoplasmic membrane [2] [1]. Unlike many other transport systems that consume ATP, the PTS is uniquely energy-efficient, utilizing phosphoenolpyruvate (PEP) as its primary phosphoryl donor, which is one of the intermediates in glycolysis [3]. This phosphorylation event serves a dual purpose: it primes the sugar for entry into metabolic pathways such as glycolysis, and it creates a charged molecule that can no longer freely diffuse out of the cell, effectively trapping the nutrient intracellularly [2] [3].
Beyond its fundamental role in carbohydrate uptake, the PTS functions as a sophisticated sensory system that integrates metabolic and transcriptional regulation [4]. The phosphorylation state of PTS components, particularly EIIACrr, serves as a key indicator of the cell's nutritional status, creating a link between metabolic flux and global regulatory networks [4]. When the influx of PTS carbohydrates is high, the phosphorylated form of EIIACrr is depleted as phosphate is diverted to sugar phosphorylation. This depletion triggers a cascade of regulatory events, including inducer exclusion where non-PTS sugar transporters are inhibited, and activation of the global transcription factor cAMP-Crp that modulates the expression of hundreds of genes [4]. This intricate coordination allows bacteria to prioritize efficiently among available carbon sources, making the PTS a central hub in bacterial metabolic control and a fascinating subject for systems biology approaches like Flux Balance Analysis.
System Architecture and Molecular Mechanism
Core Components and Phosphate Transfer Cascade
The PTS operates through a precisely coordinated phosphorylation cascade that transfers a phosphate group from phosphoenolpyruvate to the incoming sugar substrate. This process involves a series of conserved protein components that can be categorized into general and substrate-specific elements. The system's architecture ensures both efficiency and regulatory capacity, allowing bacteria to monitor and respond to their nutritional environment.
Table 1: Core Protein Components of the Phosphotransferase System
| Component | Type | Function |
|---|---|---|
| Enzyme I (EI) | General cytoplasmic protein | First component; autophosphorylates using PEP [3] [4] |
| HPr | General cytoplasmic protein | Histidine-containing protein; intermediate phosphate carrier [3] [4] |
| EIIACrr | Sugar-specific cytoplasmic protein | Key regulatory node; transfers phosphate to EIIB or binds to non-PTS transporters for inducer exclusion [4] |
| EIIB | Sugar-specific membrane-associated protein | Phosphorylates the sugar substrate during transport [3] |
| EIIC | Sugar-specific transmembrane protein | Forms the translocation channel through the membrane [3] |
The phosphate transfer follows a defined pathway: PEP → EI → HPr → EIIA → EIIB → Sugar [4]. This cascade begins when Enzyme I extracts a phosphate group from PEP and becomes autophosphorylated. The phosphate is subsequently transferred to HPr, then to the sugar-specific EIIA component, and finally to EIIB, which directly phosphorylates the sugar as it passes through the EIIC transmembrane channel [3]. The phosphorylation state of each component in this cascade, particularly EIIACrr, serves as a key indicator of the cell's metabolic status, with high glycolytic flux resulting in a lower degree of EIIACrr phosphorylation [4].
Modular Organization and Functional Domains
The PTS exhibits a modular organization that enhances its functional versatility. While the general components EI and HPr are conserved across the system, the enzyme II complexes display remarkable diversity tailored to specific carbohydrate substrates [3]. E. coli alone possesses at least 15 distinct EII complexes, each capable of transporting a different sugar or related group of sugars [3]. These EII complexes can exist as separate proteins or as fusion proteins with multiple domains strung together in a single polypeptide chain [3].
Sequence comparisons and topological studies have identified several functional domains within the PTS proteins: membrane-bound pore formation, substrate-binding sites, linker domains, transphosphorylation domains, and primary phosphorylation sites [1]. This modular architecture supports the evolution of new specificities and allows the same energy production machinery (EI and HPr) to power multiple different sugar transporters. The structural flexibility of the PTS enables it to function not only in carbohydrate transport but also as a critical component in chemotaxis and global regulatory networks [1].
Experimental Protocols for PTS Analysis
Protocol 1: Membrane Proteome Preparation for PTS Protein Quantitation
Objective: To extract and digest membrane proteins, including PTS transporters, without solubility-related bias for subsequent LC-MS/MS analysis. This protocol is essential for comprehensive quantification of PTS components and their abundance under different growth conditions [5].
Materials:
- E. coli whole cell lysate (e.g., BW25113 strain)
- Sodium deoxycholate (SDC)
- Sodium N-lauroylsarcosinate (SLS)
- Ammonium bicarbonate buffer
- Immobilized trypsin beads (e.g., Poroszyme)
- Ethyl acetate
- Trifluoroacetic acid
- Strong cation exchange (SCX) and C18 StageTips
Procedure:
- Protein Extraction: Extract proteins from E. coli whole cell lysate using a solution containing 12 mM SDC, 12 mM SLS, and 50 mM ammonium bicarbonate supplemented with a protease inhibitor such as 1 mM 4-(2-aminoethyl)benzenesulfonyl fluoride hydrochloride [5].
- Reduction and Alkylation: Reduce proteins with 10 mM dithiothreitol at room temperature for 30 minutes. Subsequently, alkylate with 55 mM iodoacetamide in the dark at room temperature for 30 minutes [5].
- Digestion: Dilute the protein mixture 5-fold with 50 mM ammonium bicarbonate. Load onto pipette tips packed with immobilized trypsin beads. Digest by centrifugation at 100 × g for 15 minutes [5].
- Surfactant Removal: Add an equal volume of ethyl acetate to the eluent and acidify with 0.5% trifluoroacetic acid (final concentration). Shake for 1 minute and centrifuge at 15,700 × g for 2 minutes. Collect the aqueous phase containing the peptides [5].
- Peptide Cleanup and Fractionation: Fractionate peptides with SCX-StageTips and desalt with C18-StageTips [5].
Applications in PTS Research: This protocol enables unbiased sample preparation, allowing protein abundance estimation based on the number of observed peptides per protein. It has been successfully used to determine copy numbers per cell for over 1,000 E. coli proteins, including 545 membrane proteins, which is crucial for constraining FBA models with quantitative proteomic data [5].
Protocol 2: Growth Rate Mapping to PTS Phosphorylation Status
Objective: To establish a relationship between the specific growth rate and the phosphorylation status of EIIACrr, the key output of the PTS sensor system [4].
Materials:
- E. coli wild-type and relevant mutant strains (e.g., ΔptsG)
- Defined media with single carbon sources (e.g., glucose, lactose, glycerol)
- Equipment for monitoring culture growth (spectrophotometer)
- Reagents for rapid protein fixation and immunoblotting
- Phospho-specific antibodies for EIIACrr or materials for Phos-tag gel electrophoresis
Procedure:
- Culture Growth: Grow E. coli strains in defined media with a single PTS (e.g., glucose) or non-PTS (e.g., lactose, glycerol) carbon source. Maintain precise environmental control (temperature, aeration) [4].
- Growth Monitoring: Monitor culture growth spectrophotometrically by measuring optical density at 600 nm (OD600) to determine the specific growth rate (μ) during mid-exponential phase.
- Sample Collection: Collect cell samples at multiple specific growth rates, which can be achieved by using different carbon sources or by nutrient limitation in chemostat cultures.
- Rapid Fixation: Rapidly fix samples to preserve the in vivo phosphorylation status of PTS proteins.
- Phosphorylation Analysis: Determine the phosphorylation ratio of EIIACrr using phospho-specific antibodies, Phos-tag SDS-PAGE, or other relevant phosphoprotein detection techniques.
Applications in PTS Research: This experimental approach generates quantitative data that can be used to parameterize and validate mathematical models of the PTS. The relationship between growth rate and EIIACrr phosphorylation provides critical insights for FBA by linking metabolic flux to regulatory signals [4].
Table 2: Research Reagent Solutions for PTS Studies
| Reagent / Material | Function / Application | Example Use Case |
|---|---|---|
| Sodium Deoxycholate (SDC) | Surfactant for unbiased membrane protein solubilization | Sample preparation for membrane proteomics [5] |
| Immobilized Trypsin | Rapid and efficient protein digestion | Proteolytic digestion for LC-MS/MS sample preparation [5] |
| Strong Cation Exchange (SCX) Tips | Peptide fractionation | Reduction of sample complexity prior to LC-MS/MS [5] |
| C18 StageTips | Peptide desalting and concentration | Sample cleanup for mass spectrometry [5] |
| Phos-tag Acrylamide | Electrophoretic separation of phosphoproteins | Determining EIIACrr phosphorylation status [4] |
Integration with Flux Balance Analysis (FBA)
Flux Balance Analysis is a constraint-based mathematical approach for analyzing the flow of metabolites through a metabolic network, particularly genome-scale metabolic reconstructions [6]. FBA calculates the flow of metabolites through this network, enabling predictions of growth rates or production of biotechnologically important metabolites without requiring detailed kinetic parameters [6]. The method is based on imposing mass balance constraints (represented by the stoichiometric matrix S, where Sv = 0 at steady state) and boundary conditions (upper and lower flux bounds) on the system, then using linear programming to find an optimal flux distribution that maximizes or minimizes a specified objective function, such as biomass production [6].
Integrating the PTS into FBA models requires special consideration of several unique features. The PTS couples transport directly to phosphorylation, meaning the traditional separation of transport and metabolic reactions in models needs refinement. Furthermore, the regulatory functions of the PTS, particularly inducer exclusion and catabolite repression mediated by EIIACrr, must be incorporated as additional constraints to accurately predict bacterial behavior in environments with multiple carbon sources [4].
Protocol 3: Constraining FBA with PTS-Derived Parameters
Objective: To incorporate PTS-related constraints and regulatory rules into a metabolic model for simulating growth on different carbon sources and predicting the effects of genetic perturbations.
Materials:
- Metabolic reconstruction of E. coli (e.g., from BiGG Models database)
- COBRA Toolbox or similar constraint-based modeling software
- Quantitative data on PTS protein abundance and phosphorylation states
- Experimental growth data for validation
Procedure:
- Model Curation: Ensure the metabolic model accurately represents all PTS transporters and their associated reactions, including the stoichiometry of PEP consumption and phosphate transfer during sugar transport [6].
- Constraint Definition: Incorporate the regulatory rules of the PTS as additional constraints:
- Inducer Exclusion: When EIIACrr is predominantly unphosphorylated (high PTS sugar flux), constrain the upper bounds of non-PTS sugar uptake reactions (e.g., lactose, glycerol) to simulate inhibition [4].
- Carbon Catabolite Repression: Link the phosphorylation state of EIIACrr to the expression of alternative metabolic genes via the cAMP-Crp complex in multi-scale models [4].
- Flvo Optimization: Set biomass production as the objective function and use linear programming to solve for the optimal flux distribution under the defined constraints [6].
- Model Validation: Compare predicted growth rates and carbon source utilization patterns with experimental data for wild-type and PTS mutant strains (e.g., ΔptsG) [4].
Application Example: This approach can simulate the classic diauxic growth of E. coli on a mixture of glucose and lactose. The model would correctly predict preferential utilization of glucose (a PTS sugar) followed by a lag phase and subsequent growth on lactose, as the relief of inducer exclusion and activation of lac operon expression occurs only after glucose depletion [4].
Table 3: FBA Predictions vs. Experimental Observations for E. coli Growth
| Condition | Carbon Source | Predicted Growth Rate (h⁻¹) | Experimental Growth Rate (h⁻¹) | Key PTS Involvement |
|---|---|---|---|---|
| Aerobic | Glucose (PTS) | 1.65 [6] | ~1.65 [6] | High glucose uptake via PTS; Low EIIA~P~ [4] |
| Anaerobic | Glucose (PTS) | 0.47 [6] | ~0.47 [6] | Reduced energy yield; Altered PTS flux [6] |
| Aerobic | Lactose (non-PTS) | Model-dependent | Measurable | EIIA~P~ inhibits lactose permease (Inducer exclusion) [4] |
| ΔptsG mutant | Glucose (PTS) | Near zero | Severely impaired | Lacks major glucose transporter PtsG [4] |
The Phosphotransferase System represents a paradigm of biological efficiency and integration, combining transport, phosphorylation, and regulatory functions in a single coordinated system. For researchers employing Flux Balance Analysis, accurately representing the PTS is crucial for predicting bacterial metabolic behavior, particularly in environments with multiple carbon sources. The experimental protocols outlined—for proteomic quantification of PTS components, determination of their phosphorylation status, and integration of PTS-derived constraints into metabolic models—provide a comprehensive framework for advancing systems-level studies of bacterial metabolism.
The multi-functional nature of the PTS makes it an attractive target for metabolic engineering and antibiotic development. Understanding the PTS through FBA can inform strategies for redirecting carbon flux in industrial biotechnology or designing compounds that disrupt this essential system in pathogenic bacteria. Future research directions include developing more sophisticated representations of PTS regulation in multi-scale models and integrating high-throughput phosphoproteomic data with constraint-based modeling to create dynamic, condition-specific models of bacterial metabolism.
The glucose-specific transporter IICBGlc, a central component of the bacterial phosphoenolpyruvate (PEP)-dependent phosphotransferase system (PTS), is responsible for the concomitant uptake and phosphorylation of glucose in Escherichia coli and many other bacteria [7] [8]. Recent advances in single-particle cryogenic-electron microscopy (cryo-EM) have elucidated its high-resolution structure and the molecular details of its unique transport mechanism. This transporter employs an elevator-type mechanism, where a mobile transport domain undergoes large-scale rigid-body movement to shuttle the substrate across the membrane relative to a static scaffold domain [7] [9]. Historically, structures of such transporters were captured only in inward-facing (IF) or outward-facing (OF) conformations. However, a landmark study has now trapped IICBGlc in a previously unobserved intermediate state, stalled by a detergent molecule bound to the substrate site [7] [9]. This breakthrough provides unprecedented insights into the transport cycle and reveals a novel stalling mechanism, paving the way for the rational design of specific inhibitors that could target this essential bacterial nutrient uptake system [7].
Table 1: Key Structural Determinations of the IICBGlc Transporter
| PDB/EMDB ID | Resolution | Conformational State | Bound Ligand(s) | Key Findings | Citation |
|---|---|---|---|---|---|
| 8QST / EMD-18641 | 2.89 Å | Occluded Inward- & Outward-Facing | Glucose (BGC) | Revealed the endpoints of the elevator cycle in a lipid environment. [10] | |
| 9HNP / EMD-52311 | 2.53 Å | Intermediate State | n-dodecyl-β-D-maltopyranoside (LMT) | Captured the transport domain halfway, revealing a stalling mechanism. [9] |
The phosphotransferase system (PTS) is a major carbohydrate uptake system in bacteria, structurally coupling the transmembrane transport of sugars with their phosphorylation using phosphoenolpyruvate (PEP) as the phosphate donor [8] [11]. The glucose-specific PTS in E. coli involves three soluble cytoplasmic proteins—Enzyme I (EI), HPr, and IIAGlc—and the membrane-bound transporter IICBGlc, which is encoded by the ptsG gene [8]. The IICBGlc protein itself is bifunctional: the transmembrane IICGlc domain is responsible for glucose binding and translocation, while the cytoplasmic IIBGlc domain phosphorylates the transported glucose using a phosphate group received from IIAGlc [7] [11]. The final product released into the cytoplasm is glucose-6-phosphate, a glycolytic intermediate [7].
Beyond its primary metabolic role, IICBGlc plays a critical regulatory function. The phosphorylation state of IICBGlc, determined by the availability of external glucose, governs its interaction with a global transcriptional repressor, Mlc [8] [12]. When glucose is present, IICBGlc is dephosphorylated and sequesters Mlc to the membrane, thereby derepressing genes involved in sugar metabolism, including the ptsG and ptsHIcrr operons [8] [12]. This dual role as a transporter and a regulatory node underscores the central importance of IICBGlc in bacterial physiology.
Structural Architecture and the Elevator Mechanism
The IICGlc domain forms a stable dimer, with each monomer consisting of two distinct structural units: a scaffold domain and a transport domain (TD) [7] [9]. The scaffold domain is involved in dimer formation and remains static within the membrane. The transport domain, which contains the sugar-binding site, is highly mobile [7].
Transport is mediated by an elevator-type mechanism, where the entire transport domain undergoes a large-scale, rigid-body movement, sliding up and down relative to the static scaffold domain like a elevator car [7] [9]. This motion translocates the substrate-binding site from one side of the membrane to the other.
Table 2: Conformational States of the IICGlc Transport Domain
| Conformational State | Position of Transport Domain | Accessibility of Substrate Site | Key Structural Features |
|---|---|---|---|
| Outward-Facing (OF) | Positioned towards the periplasm | Accessible from the extracellular side | Occluded state with bound glucose has been observed. [10] |
| Intermediate State | Halfway between OF and IF | Not accessible from either side | TD movement is stalled; observed with bulky DDM ligand. [9] |
| Inward-Facing (IF) | Positioned towards the cytoplasm | Accessible from the cytoplasmic side | Occluded state with bound glucose has been observed. [10] |
The recent cryo-EM structure (PDB: 9HNP) captured IICGlc in an intermediate conformation, where the transport domain is frozen halfway through its trajectory [9]. This was achieved by the binding of the detergent n-dodecyl-β-D-maltopyranoside (DDM) to the glucose-binding site. The bulky nature of DDM, coupled with a shift in the "thin gate" of the transporter, physically impedes the sliding of the transport domain, thereby stalling the transport cycle [7] [9]. This structure provides a direct snapshot of the elevator mechanism in action and reveals the molecular determinants that could be targeted to inhibit the transporter.
Diagram 1: The IICBGlc Transport and Phosphorylation Cycle. This workflow illustrates the elevator mechanism, highlighting the newly discovered intermediate state.
Detailed Experimental Protocols
Cryo-EM Sample Preparation and Data Collection for IICBGlc
Objective: To determine the high-resolution structure of the E. coli IICBGlc transporter in a native-like lipid environment and in a detergent-solubilized, stalled state.
Materials:
- E. coli strain expressing recombinant IICBGlc (e.g., from plasmid pRE1-ptsG) [8].
- Detergents: n-Dodecyl-β-D-maltopyranoside (DDM) for solubilization and structure determination [7] [9].
- Lipids for reconstitution into lipid nanodiscs (for native-like structures) [10].
- Cryo-EM grids (e.g., Quantifoil R1.2/1.3 or UltrAuFoil).
- Vitrification device (e.g., Vitrobot Mark IV).
Methodology:
- Protein Expression and Purification: IICBGlc is overexpressed in E. coli and membranes are isolated. The transporter is solubilized from the membranes using DDM and purified via affinity and size-exclusion chromatography (SEC) [7] [9].
- Grid Preparation and Vitrification:
- Cryo-EM Data Collection and Processing:
- High-resolution micrographs are collected on a high-end cryo-EM (e.g., Titan Krios) equipped with a high-quality detector [10] [9].
- Data processing is performed in software suites like cryoSPARC [10] [9]:
- Patch motion correction and CTF estimation.
- Particle picking, extraction, and 2D classification.
- Ab-initio reconstruction and heterogeneous refinement to separate conformational states.
- Non-uniform refinement to achieve high-resolution 3D reconstructions.
- Model Building and Refinement: An initial model is built de novo or based on a related structure and is iteratively refined against the cryo-EM map using programs like PHENIX [10] [9]. The model is validated for geometry and fit to the density.
Functional Analysis:In VivoTransport and Regulation Assays
Objective: To validate the physiological function of IICBGlc and its regulatory interaction with Mlc.
Materials:
- E. coli wild-type and mutant strains (e.g., ΔptsG, Δmlc, ΔptsI) [8] [12].
- Radiolabeled glucose (e.g., ¹⁴C-glucose) for transport assays.
- Antibodies against Mlc for western blotting and co-precipitation [12].
- PEP and soluble PTS proteins (EI, HPr, IIAGlc) for in vitro phosphorylation assays [8].
Methodology:
- Mlc Localization Assay:
- Cultivate E. coli cells in media with or without glucose.
- Separate cytoplasmic and membrane fractions by ultracentrifugation.
- Analyze the distribution of Mlc in both fractions by western blotting. Expect Mlc to be membrane-associated in the presence of glucose (dephosphorylated IICBGlc) and cytoplasmic in its absence (phosphorylated IICBGlc) [12].
- Co-precipitation Assay:
- Incubate purified Mlc with IICBGlc-enriched membrane vesicles.
- Phosphorylate the PTS components by adding PEP, EI, HPr, and IIAGlc. Use glucose to promote dephosphorylation.
- Pellet the membranes. Analyze the supernatant and pellet for the presence of Mlc. Mlc co-pellets with membranes only when IICBGlc is dephosphorylated (glucose condition) [8].
- In Vivo Transcription Assay:
- Isolate total RNA from strains overproducing or lacking IICBGlc.
- Perform primer extension or S1 nuclease analysis using probes specific for Mlc-regulated promoters (e.g., ptsG or ptsHIcrr P0). Overproduction of IICBGlc should lead to derepression (induction) of these promoters even in the absence of glucose [8].
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Research Reagents for IICBGlc and PTS Studies
| Reagent / Material | Function / Role in Experimentation | Example Use Case |
|---|---|---|
| n-Dodecyl-β-D-maltopyranoside (DDM) | Mild detergent for solubilizing and stabilizing membrane proteins. Also acts as a stalling ligand in structural studies. | Used to solubilize IICBGlc and serendipitously trap its intermediate conformation for cryo-EM [7] [9]. |
| Lipid Nanodiscs (e.g., MSP-based) | Membrane mimetic system that provides a native-like lipid bilayer environment for structural and functional studies. | Used for determining the inward- and outward-facing structures of IICBGlc under near-native conditions [10]. |
| Phosphoenolpyruvate (PEP) | Phosphate donor for the PTS phosphorylation cascade. | Used in in vitro assays to establish the phosphorylated state of PTS components, including IICBGlc [8]. |
| Anti-Mlc Antibody | Specific detection and immunoprecipitation of the Mlc repressor protein. | Used in western blotting and co-precipitation assays to study the IICBGlc-Mlc interaction and cellular localization of Mlc [12]. |
| cryoSPARC Software Suite | Comprehensive software for processing single-particle cryo-EM data. | Used for 2D classification, 3D reconstruction, and refinement of IICBGlc cryo-EM structures [10] [9]. |
Regulatory Signaling and Research Applications
The structural and functional insights into IICBGlc have profound implications for understanding bacterial physiology and for applied drug discovery. The regulatory circuit involving IICBGlc and Mlc is a key model for transcriptional regulation in response to nutrient availability.
Diagram 2: The IICBGlc-Mlc Regulatory Signaling Pathway. This diagram shows how external glucose controls gene expression via the phosphorylation state of IICBGlc.
The discovery of the intermediate, stalled conformation of IICBGlc directly enables structure-based inhibitor design. Bulky molecules that mimic the stalling effect of DDM could be developed into specific anti-bacterial compounds [7] [9]. Given that the PTS is present in many pathogenic bacteria but absent in humans, IICBGlc represents an excellent, selective drug target. Furthermore, a detailed understanding of the elevator mechanism and its regulation contributes to the fundamental field of membrane transport biology, providing a model for studying other elevator-type transporters found in all domains of life.
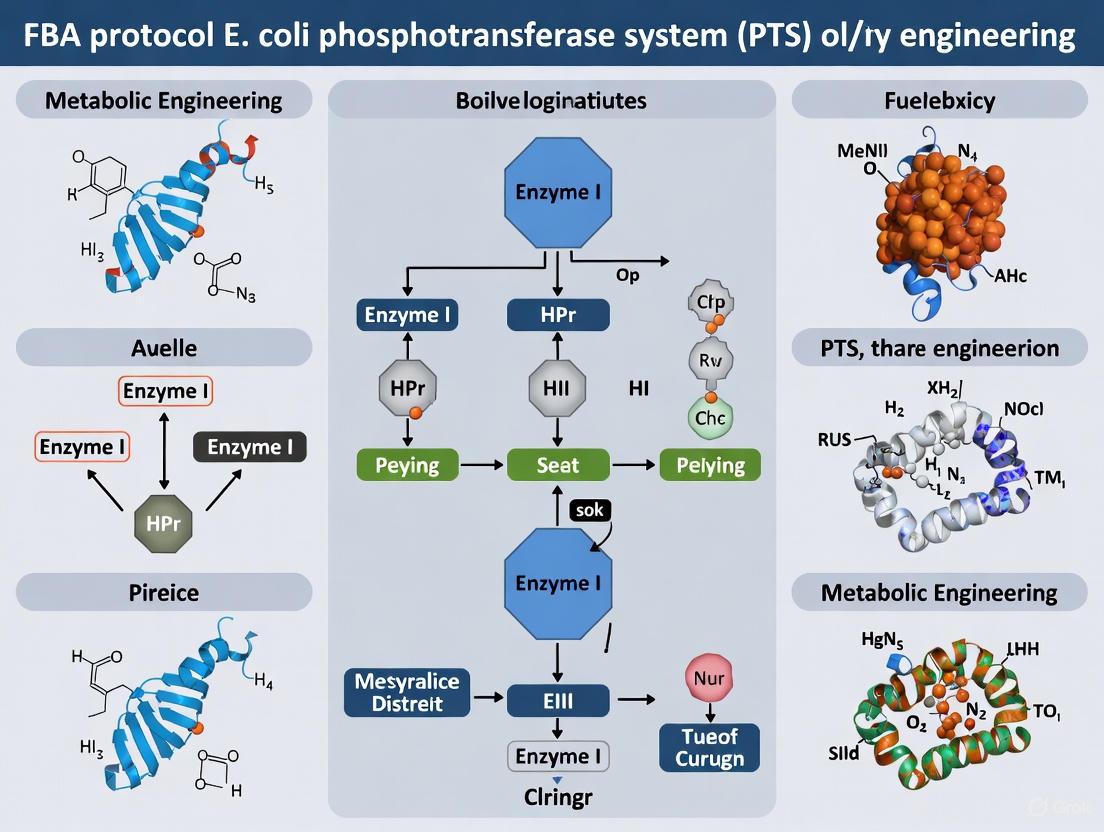
The phosphoenolpyruvate (PEP):carbohydrate phosphotransferase system (PTS) is a multifaceted biological system found exclusively in bacteria, responsible for the transport and concomitant phosphorylation of a wide range of sugar substrates, including glucose, fructose, mannose, and mannitol [13] [14]. This system couples the energy derived from the glycolytic intermediate PEP to the vectorial phosphorylation of sugars during their translocation across the cell membrane, resulting in the intracellular accumulation of sugar-phosphates like glucose-6-phosphate, which are direct substrates for glycolysis [15] [14]. The PTS employs a phosphorylation cascade, a "bucket-brigade" style transfer of a phosphoryl group through several protein components, which culminates in the modification of the incoming sugar [16] [14]. Beyond its primary metabolic role, the phosphorylation status of PTS components serves as a key regulatory node, controlling carbohydrate uptake, gene expression, enzyme activity, and even bacterial virulence [13]. This document details the mechanism, experimental analysis, and integration of the PTS within the framework of Flux Balance Analysis (FBA) for E. coli research.
Core Mechanism of the Phosphotransfer Cascade
The PTS is composed of general cytoplasmic proteins and sugar-specific enzyme complexes. The phosphoryl group from PEP is transferred via a series of proteins to the incoming sugar. The sequence of the phosphorylation cascade for a glucose-like substrate is as follows:
- Enzyme I (EI): The phosphoryl group from PEP is transferred to a conserved histidine residue on EI. This step requires Mg²⁺ and is common to the uptake of all PTS sugars [14].
- Histidine-containing Phosphocarrier Protein (HPr): The phosphoryl group is then transferred from EI to a histidine residue on HPr [14].
- Sugar-specific Enzyme IIA (EIIA): The phosphoryl group is passed from HPr to a histidine residue on a sugar-specific EIIA component. In the case of glucose in enteric bacteria, this protein is IIAGlc [14].
- Sugar-specific Enzyme IIB (EIIB): The phosphoryl group is transferred from EIIA to a cysteine or histidine residue on the membrane-associated EIIB component [14].
- Sugar Translocation and Phosphorylation via Enzyme IIC (EIIC): The phosphoryl group is finally transferred from EIIB to the sugar substrate as it is translocated across the membrane through the integral membrane transporter, EIIC. The transported sugar, such as glucose, is thereby phosphorylated to glucose-6-phosphate upon entry into the cytoplasm [15] [14].
This cascade creates a repressive interaction between different PTS sugars. Multiple sugars compete for the limited phosphoryl flux from PEP and shared early components (EI and HPr). This competition, combined with transcriptional feedback, results in a "winner-take-all" dynamic that underlies phenomena like diauxic growth, where bacteria consume preferred sugars sequentially rather than simultaneously [16].
Visualizing the Phosphotransfer Cascade and Transport
The following diagram illustrates the complete phosphorylation cascade and the accompanying transport mechanism.
Diagram 1: The PTS phosphorylation cascade for glucose. The phosphoryl group (P) from PEP is transferred sequentially via the general proteins EI and HPr to the sugar-specific components EIIAGlc and EIIBGlc. EIIBGlc phosphorylates glucose as it is transported across the membrane by the EIICGlc permease, producing intracellular glucose-6-phosphate. The EIICGlc transporter operates via an elevator-type mechanism, shifting its transport domain to present the substrate-binding site to either side of the membrane [15].
Application Notes & Experimental Protocols
Protocol: Measuring PTS Function via Intracellular Energy Status
The functional state of the PTS is intimately linked to the cellular energy charge. This protocol outlines a method to quantify the intracellular [ATP]/[ADP] ratio, a key metric of energy status, in E. coli strains with perturbed PTS function, such as a PTS- mutant (e.g., PB12) [17].
1. Objective: To determine the impact of PTS disruption on the intracellular energy charge by measuring the [ATP]/[ADP] ratio.
2. Materials:
- E. coli strains: Wild-type (e.g., JM101) and PTS- mutant (e.g., PB12).
- M9 minimal medium with a defined carbon source (e.g., 2 g/L glucose).
- Lysis buffer.
- ENLITEN ATP Assay System (Promega) or equivalent.
- Pyruvate Kinase (PK) from Sigma-Aldrich.
- Phosphoenolpyruvate (PEP).
- Luminometer.
- Cyclic AMP Enzyme Immunoassay Kit (for concomitant cAMP analysis).
3. Methodology: 1. Culture Conditions: Grow biological quadruplicates of each strain in 1L bioreactors with controlled conditions: M9 medium, 2 g/L glucose, pH 7.0 (maintained with NH₄OH), 37°C, 600 rpm agitation, and 1 vvm airflow [17]. 2. Sample Harvesting: Withdraw 50 mL culture samples during the mid-exponential growth phase (OD₆₀₀ ≈ 1.0). Rapid cooling and centrifugation are critical to preserve metabolite levels. 3. Cell Lysis: Lyse the cell pellets using a standardized method, such as cold acid extraction or bead beating, to release intracellular nucleotides [17]. 4. ATP Determination: Use the luminescence-based ATP assay according to the manufacturer's instructions to determine the ATP concentration in the cell lysate. 5. ADP Determination: Add 1 U of Pyruvate Kinase and 1 mM PEP to an aliquot of the same lysate. This enzymatically converts ADP to ATP. Measure the resulting increase in luminescence, which corresponds to the original ADP concentration [17]. 6. Data Calculation: Calculate the [ATP]/[ADP] ratio using the corrected values for ATP and ADP concentrations.
4. Data Interpretation:
- A lower [ATP]/[ADP] ratio in the PTS- mutant compared to the wild-type indicates a disruption in energy metabolism, consistent with the loss of the high-efficiency PTS uptake system [17].
- This energetic perturbation can be linked to transcriptional changes in central carbon metabolism genes observed via RT-qPCR, providing a integrated view of the metabolic response [17].
5. Integration with FBA:
- The measured [ATP]/[ADP] ratio can be used as a constraint in a genome-scale metabolic model of E. coli.
- By imposing the experimentally determined ratio, the FBA solution space is refined, leading to more accurate predictions of flux distributions through the PTS and other metabolic pathways under the specific genetic perturbation.
Quantitative Data from PTS and Energy Perturbation Studies
The following table summarizes key physiological and metabolic data from studies investigating PTS mutants and energy perturbations in E. coli.
Table 1: Physiological and Transcriptional Response of E. coli to PTS and Energy Perturbation [17].
| Strain / Condition | Specific Glucose Uptake Rate, qGlc (mmol/gDCW/h) | [ATP]/[ADP] Ratio | Intracellular cAMP Level | Key Transcriptional Responses |
|---|---|---|---|---|
| Wild-type (JM101) | Baseline | High | Low | Standard expression of glycolytic and TCA cycle genes. |
| PTS- Mutant (PB12) | Increased vs. Wild-type | Reduced vs. Wild-type | Elevated vs. Wild-type | Upregulation of non-PTS transporters (e.g., GalP), TCA cycle, glyoxylate shunt, and respiratory genes. |
| PTS- with F1-ATPase Overexpression (PB12AGD+) | Further Increased | Severely Reduced | Reduced vs. PB12 | Carbon flux redirected toward PPP and Entner-Duodoroff pathways; further upregulation of respiratory genes. |
Abbreviations: DCW: Dry Cell Weight; PPP: Pentose Phosphate Pathway; TCA: Tricarboxylic Acid Cycle.
Protocol: Computational Analysis of PTS using Flux Balance Analysis
FBA is a powerful constraint-based modeling approach used to predict metabolic flux distributions in a genome-scale metabolic network. This protocol details its application to study the PTS in E. coli.
1. Objective: To predict the growth rate and internal flux distribution of E. coli grown on different PTS sugars and to identify optimal metabolic strategies.
2. Materials:
- A genome-scale metabolic model of E. coli (e.g., iJO1366).
- Constraint-based modeling software (e.g., CobraPy in Python, the COBRA Toolbox for MATLAB).
- Sugar uptake rates (experimentally determined or theoretically set).
3. Methodology:
1. Model Construction and Curation: Ensure the metabolic model accurately represents the PTS reactions for the relevant sugars (e.g., glucose, mannose). This includes the transport reaction (e.g., glc-D[e] -> g6p[c]) and the associated energy expenditure from PEP.
2. Defining Constraints:
- Set the lower and upper bounds for the sugar uptake reaction based on experimental data or to a fixed value (e.g., -10 mmol/gDW/h).
- Set the oxygen uptake rate to simulate aerobic or anaerobic conditions.
- Optionally, constrain the flux through other reactions based on gene knockout data (e.g., set PTS enzyme fluxes to zero for a PTS- mutant simulation).
3. Defining the Objective Function: Typically, biomass production is set as the objective function to be maximized, simulating the evolutionary pressure for growth.
4. Solving the Linear Programming Problem: Use the optimization algorithm to find the flux distribution that maximizes the objective function, given the network stoichiometry and constraints.
4. Data Interpretation:
- The model will predict a maximal growth rate and a full flux map.
- In a mixture of two PTS sugars, an optimal strategy often involves a discontinuous switch where the preferred sugar (e.g., the one with higher ζjSj/αj, representing energy yield and synthesis cost) is exclusively consumed until depletion, followed by a metabolic lag and then consumption of the second sugar—a prediction that matches observed diauxic growth [16].
- FBA can reveal how competition for limited phosphoryl flux through shared PTS components (EI, HPr) creates a "winner-take-all" dynamic, explaining catabolite repression [16].
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Resources for PTS Research.
| Reagent / Resource | Function / Application | Examples & Notes |
|---|---|---|
| Genetically Modified Strains | To study the function of specific PTS components via gene deletion or overexpression. | E. coli PB12 (ΔptsHIcrr); BW25113ΔacrB for membrane protein purification [17] [18]. |
| Plasmid Vectors | For heterologous expression and purification of PTS proteins. | pTrc99A vector with inducible trc promoter [17]. |
| ATP/ADP Assay Kits | To quantify intracellular energy charge and monitor PTS activity. | ENLITEN ATP Assay System (Promega); coupled enzyme assay with Pyruvate Kinase for ADP [17]. |
| cAMP Immunoassay Kits | To measure intracellular cAMP levels, a key indicator of catabolite repression status. | Cyclic AMP Enzyme Immunoassay Kit; levels are inversely related to IIAGlc phosphorylation [17] [14]. |
| Lipid Nanodiscs | To reconstitute purified membrane transporters like EIICGlc for structural and biophysical studies in a native-like lipid environment. | Used for cryo-EM structure determination of IICBGlc [15]. |
| Genome-Scale Metabolic Models | For computational analysis of PTS and metabolic networks using FBA. | E. coli model iJO1366; constraints can be adjusted to simulate PTS mutants [16]. |
| Pathway Modeling Tools | To create standardized, reusable visual models of the PTS pathway. | PathVisio, CellDesigner; use Systems Biology Graphical Notation (SBGN) [19]. |
The complete phosphotransferase system in Escherichia coli
The phosphoenolpyruvate (PEP)-dependent phosphotransferase system (PTS) in Escherichia coli represents a sophisticated multiprotein system that performs dual physiological roles: (i) the active transport and phosphorylation of numerous sugars, and (ii) global regulatory control that coordinates carbon metabolism with other cellular processes. A critical understanding for researchers applying Flux Balance Analysis (FBA) is that the PTS is not a single entity but comprises two distinct branches: the well-characterized sugar-transport PTS and the regulatory PTSNtr (Nitrogen-related PTS). These branches share structural similarities in their phosphoryl transfer chain but serve fundamentally different biological functions. This application note delineates the components, functions, and interactions of these two systems, providing a structured framework for incorporating their biochemical constraints into metabolic models such as FBA simulations. The PTS accounts for the uptake of numerous carbohydrates in E. coli, simultaneously transporting and phosphoryating its substrates using PEP as the phosphoryl donor. Beyond this primary metabolic role, the phosphorylation state of PTS components serves as a global sensory network, regulating processes including catabolite repression, inducer exclusion, and gene expression.
Table 1: Core Components of the Two PTS Branches in E. coli
| Component Type | Sugar-Transport PTS | Regulatory PTSNtr |
|---|---|---|
| General Proteins | Enzyme I (EI), HPr | Enzyme INtr (EINtr), NPr |
| Role of General Proteins | Phosphoryl transfer from PEP to EIIA components | Phosphoryl transfer from PEP to PtsN (EIIANtr) |
| Key Specific Protein | Various EIIA/EIIB/EIIC complexes (sugar-specific) | PtsN (EIIANtr) |
| Primary Function | Sugar uptake and phosphorylation | Regulatory signaling, K+ homeostasis |
| Genetic Locus | ptsH, ptsI, crr operon [20] |
rpoN operon (ptsP, ptsO, ptsN) [21] |
System Architecture and Key Differentiators
The Sugar-Transport PTS: A Major Carbohydrate Uptake System
The sugar-transport PTS is composed of a phosphorylation cascade that initiates with the general, non-sugar-specific proteins Enzyme I (EI) and the histidine phosphocarrier protein (HPr). The phosphoryl group from PEP is sequentially transferred to EI, then to HPr, and subsequently to sugar-specific enzymes II (EII complexes) [18]. Each EII complex is typically comprised of three domains or subunits: IIA (membrane-associated, energy-coupling), IIB (cytoplasmic, phosphorylating), and IIC (membrane-embedded, transport). A comprehensive genomic analysis has identified 21 (and possibly 22) distinct Enzyme II complexes in E. coli, which are phylogenetically categorized into families, primarily the fructose (Fru), glucose (Glc), and other permease families [22]. For instance, the glucose-specific transporter is a member of the Glucose-Fructose-Lactose (GFL) superfamily and consists of the soluble IIAGlc protein and the membrane protein IICBGlc [18] [15]. A 2024 cryo-EM structural study revealed that the IICGlc domain functions as an elevator-type transporter, with substantial rigid-body movement to alternate the substrate-binding site between the periplasmic and cytoplasmic sides of the membrane [15]. Another well-characterized example is the D-sorbitol PTS permease, encoded by the srlABE genes (EC 2.7.1.198), which catalyzes the phosphorylation of D-sorbitol to D-sorbitol 6-phosphate during transport [23].
The Regulatory PTSNtr: A Signal Transduction Pathway
In contrast to the sugar-transport PTS, the PTSNtr lacks a permease component and is therefore not involved in sugar uptake [21]. Its phosphoryl transfer chain is genetically encoded within the rpoN and ptsP operons [22] and consists of:
- Enzyme INtr (EINtr, ptsP)
- NPr (ptsO)
- EIIANtr (ptsN)
The phosphate flow proceeds from PEP → EINtr → NPr → EIIANtr (PtsN). The primary role of this system is regulatory. The phosphorylation state of EIIANtr (PtsN) serves as a signal that reflects the metabolic status of the cell, influencing processes such as potassium homeostasis and potentially coordinating carbon and nitrogen metabolism [21]. Experimental work in Pseudomonas putida (which possesses a homologous system) suggests that the phosphorylation state of PtsN is influenced by the available carbon source and growth phase, acting as a "physiological reporter" of the cell's status [21].
Cross-Talk and Integrated Regulation
A crucial layer of complexity for systems biology models is the documented cross-talk between the two PTS branches. Under specific metabolic conditions, phosphoryl groups can be transferred from the sugar-transport branch to the regulatory branch. Specifically, the HPr domain of the fructose-specific PTS protein (FruB) can directly phosphorylate PtsN (EIIANtr), thereby integrating information about the nature of the degraded sugar directly into the phosphorylation state of this key regulatory node [21]. This cross-talk provides a direct mechanistic link between sugar metabolism and global regulatory outputs.
Table 2: Functional Comparison of the Sugar-Transport PTS and PTSNtr
| Feature | Sugar-Transport PTS | PTSNtr |
|---|---|---|
| Primary Physiological Role | Catalytic: Sugar uptake & phosphorylation | Regulatory: Signal transduction |
| Permease Component | Present (IIC domain) | Absent |
| Key Output | Phosphorylated sugar (e.g., Glucose-6-P) | Phosphorylated PtsN (regulatory signal) |
| Impact on FBA Models | Directly defines substrate uptake rate & growth yield | Indirect; modulates reaction bounds via regulation |
| Cross-Talk | Can donate phosphate to PTSNtr via HPr (FruB) | Can accept phosphate from sugar-transport PTS |
| Representative Components | IIAGlc, IICBGlc, SrlABE (sorbitol) | PtsP (EINtr), PtsO (NPr), PtsN (EIIANtr) |
Experimental Protocols for PTS Analysis
Protocol 1: In Vivo Analysis of PTS Operon Expression
This protocol is adapted from studies investigating the positive regulation of the pts operon (ptsH, ptsI, crr) in response to glucose [20].
1. Principle: To characterize the transcriptional regulation of the sugar-transport PTS operon using gene fusions with a reporter gene (e.g., lacZ).
2. Key Reagents:
- Strains: E. coli strains carrying
ptsH-ptsI-lacZorptsI-crr-lacZoperon fusions. - Media: Minimal media with defined carbon sources (e.g., glycerol, glucose).
- Reagents: CAP-cAMP, reagents for β-galactosidase assay (e.g., ONPG). 3. Procedure:
- Step 1: Grow the reporter strains in minimal media with a non-PTS carbon source (e.g., glycerol) to mid-exponential phase.
- Step 2: Induce the system by adding glucose or other PTS substrates to the culture.
- Step 3: Measure the expression of the operon fusions by assaying for β-galactosidase activity at regular intervals.
- Step 4: To dissect the regulatory mechanism, test the effect of overproducing EIIGlc in the absence of transport, and examine the dependence on CAP-cAMP. 4. Data Interpretation: Transcriptional stimulation by glucose is dependent on transport through EIIGlc. This regulation is mediated by an increase in the level of unphosphorylated EIIGlc, which acts as a signal during glucose transport [20].
Protocol 2: Phosphorylation State Analysis of PTSNtr Proteins
This protocol is based on methods used to study the PTSNtr in P. putida, which is highly analogous to the E. coli system [21].
1. Principle: To monitor the in vivo phosphorylation state of PtsN under different environmental conditions using a gel-shift assay. 2. Key Reagents:
- Strains: Wild-type and isogenic mutants in
ptsP,ptsO, andptsN. - Media: Minimal media with different carbon and nitrogen sources (e.g., casamino acids, CAA + fructose, CAA + glucose, ammonia, nitrate).
- Reagents: Antibodies specific to PtsN, reagents for Western blotting. 3. Procedure:
- Step 1: Grow bacterial strains under the conditions of interest and harvest cells at different growth phases (exponential vs. stationary).
- Step 2: Rapidly lyse cells using a method that preserves the phosphorylation state, such as dielectric breakdown (e.g., using a Bio-Rad Gene Pulser).
- Step 3: Separate proteins by SDS-PAGE. The phosphorylated and unphosphorylated forms of PtsN will migrate to different positions due to the altered charge and mass.
- Step 4: Perform Western blotting using anti-PtsN antibodies to visualize the ratio of phosphorylated to unphosphorylated PtsN. 4. Data Interpretation: The phosphorylation state of PtsN is dynamic, influenced by carbon source, nitrogen source, and growth stage. Non-phosphorylated PtsN predominates during rapid growth, while the phosphorylated form accumulates in the stationary phase [21].
The Scientist's Toolkit: Key Research Reagents
Table 3: Essential Research Reagents for E. coli PTS Studies
| Reagent / Material | Function / Description | Example Application |
|---|---|---|
| Anti-PtsN Antibodies | Immunodetection of PtsN and its phosphorylated forms | Western blot analysis of PtsN phosphorylation state [21] |
| pts Operon Reporter Strains | Strains with lacZ fused to pts promoters |
Quantifying transcriptional regulation of the PTS operon [20] |
| Lipid Nanodiscs | Native-like membrane mimetic for structural studies | Cryo-EM structure determination of IICBGlc [15] |
| PTS Gene Knockout Mutants | Isogenic strains with deletions in ptsH, ptsI, ptsP, ptsN, etc. |
Functional dissection of individual PTS components [21] |
| Defined Minimal Media | Media with specific PTS/non-PTS carbon sources | Controlling PTS activity and studying regulatory responses [20] [21] |
Visualization of PTS Pathways and Networks
Diagram 1: Phosphoryl Transfer in the Glucose PTS. The cascade from PEP to incoming glucose via soluble and membrane-bound components.
Diagram 2: PTSNtr Phosphoryl Transfer and Cross-Talk. The regulatory PTSNtr pathway and its interaction with the sugar-transport PTS via HPr.
Integration with Flux Balance Analysis (FBA) Protocols
Integrating the biochemical reality of the PTS into FBA models is critical for accurate simulations of E. coli growth on different carbon sources. FBA is a constraint-based mathematical approach that analyzes the flow of metabolites through a genome-scale metabolic network to predict optimal growth rates or metabolite production [6]. Key integration points include:
- Reaction Stoichiometry: The transport of a PTS sugar (e.g., glucose) must be modeled as a coupled import and phosphorylation reaction. For example:
glucose[ext] + PEP → glucose-6-phosphate + pyruvate. This stoichiometry directly links substrate uptake to PEP consumption and pyruvate production. - Energy Considerations: PTS-mediated transport does not consume ATP directly but uses PEP as an energy donor. This has distinct energetic implications compared to ATP-driven ABC transporters or proton symporters, which must be correctly represented in the model's objective function.
- Regulatory Constraints: The PTSNtr is not directly part of the metabolic network but influences it through regulation. FBA models typically do not dynamically incorporate such regulation. A practical workaround is to run separate FBA simulations under different "regimes" (e.g., simulating low PtsN~P conditions by adjusting the bounds of reactions it influences, such as potassium transport [21] or specific catabolic pathways).
- Cross-Talk Implications: The phosphate exchange between the PTS branches means that the energetic state of the two systems is linked. During growth on a PTS sugar like fructose, a significant portion of the phosphoryl groups for PtsN phosphorylation may be provided by the sugar-transport branch [21]. This interplay can be conceptually incorporated but is challenging to model quantitatively without kinetic parameters.
By accurately representing the components and constraints of both PTS branches, researchers can significantly enhance the predictive power of FBA models for simulating E. coli metabolism under a wide range of nutritional conditions.
The phosphotransferase system (PTS) is a multifaceted bacterial system that catalyzes the transport and phosphorylation of numerous carbohydrates while simultaneously regulating cellular processes in response to carbohydrate availability [24]. For researchers using genome-scale metabolic models (GSMMs) to study E. coli physiology, accurately integrating the PTS is crucial for predicting substrate utilization, growth rates, and metabolic flux distributions. The PTS represents a critical link between extracellular conditions and intracellular metabolic states. This protocol details the key reactions, stoichiometry, and methodologies required to incorporate the PTS into a GSMM, framed within a broader flux balance analysis (FBA) workflow for E. coli research.
Background: The PTS Phosphorylation Cascade
The PTS uses phosphoenolpyruvate (PEP) as an energy source and phosphoryl donor. The phosphoryl group is transferred through a series of proteins to the incoming sugar [24]. For glucose, the generally accepted phosphorylation cascade is as follows:
- Phosphoenolpyruvate (PEP) + Enzyme I (EI) → Pyruvate + Phospho-EI
- Phospho-EI + Histidine-containing Phosphocarrier Protein (HPr) → EI + Phospho-HPr
- Phospho-HPr + EIIAGlc → HPr + Phospho-EIIAGlc
- Phospho-EIIAGlc + EIIBCGlc → EIIAGlc + Phospho-EIIBCGlc
- Phospho-EIIBCGlc + Glucose → EIIBCGlc + Glucose-6-Phosphate
The overall reaction is: PEP + Glucose → Pyruvate + Glucose-6-Phosphate
This process is notable for its stoichiometric consumption of PEP and production of pyruvate during carbohydrate uptake, directly linking transport to central carbon metabolism. The PTS comprises two general cytoplasmic components, EI and HPr, and numerous sugar-specific enzyme II (EII) complexes. E. coli possesses at least 15 different EII complexes, enabling the transport of a wide variety of sugars [24].
Key PTS Reactions and Stoichiometry for GSMMs
Integrating the PTS into a GSMM requires capturing both the transport reaction and its associated energy and redox balances. The following table summarizes the core PTS transport reactions for key carbohydrates in E. coli.
Table 1: Key PTS Carbohydrate Transport Reactions and Stoichiometry in E. coli
| Carbohydrate | PTS Transport Reaction | Stoichiometric Notes |
|---|---|---|
| Glucose | pep[c] + glc-D[e] -> pyr[c] + g6p[c] |
Consumes 1 PEP, produces 1 Pyruvate per glucose. |
| Mannose | pep[c] + mann[e] -> pyr[c] + mann6p[c] |
Shares components with the glucose PTS. |
| Fructose | pep[c] + fru[e] -> pyr[c] + f1p[c] |
|
| N-Acetylglucosamine (GlcNAc) | pep[c] + gam6p[e] -> pyr[c] + gam6p[c] |
GlcNAc is a component of bacterial cell walls [25]. |
Beyond the specific transport reactions, the PTS must be integrated with the biomass objective function. The PTS impacts the synthesis of key biomass precursors. For instance, N-Acetylglucosamine-1-phosphate (GlcNAc-1-P), a PTS-related metabolite, is a vital precursor for peptidoglycan biosynthesis in E. coli [25].
Table 2: Metabolic Genes and Proteins Essential for PTS Integration
| Gene | Protein | Functional Role in PTS |
|---|---|---|
ptsI |
Enzyme I (EI) | First general PTS component; phosphorylated by PEP. |
ptsH |
HPr | Histidine-containing phosphocarrier protein. |
crr |
EIIAGlc | Glucose-specific component; central to regulatory functions. |
ptsG |
EIIBCGlc | Glucose-specific membrane transporter/permease. |
Protocol: Integrating the PTS into an E. coli GSMM
This protocol outlines the process of building a GSMM from a genome sequence and highlights the critical steps for incorporating the PTS, based on established tools like PyFBA and the COBRA Toolbox [26] [27].
Genome Annotation and Reaction Identification
The first step is identifying all metabolic genes, including those of the PTS.
- Obtain Genome Annotation: Use an annotation system like RAST or PROKKA to identify protein-encoding genes and assign functional roles [26] [27].
- Convert Functional Roles to Reactions: Map the annotated genes to enzyme complexes and then to biochemical reactions. This can be achieved using databases like Model SEED or KEGG.
- Critical Step for PTS: The PTS involves multi-protein complexes. For example, the
ptsIgene (encoding EI) is involved in several complexes associated with the import of different sugars [26]. Ensure the gene-protein-reaction (GPR) associations correctly reflect that the general components (EI, HPr) are required for all PTS sugars, while specific EII complexes are required for their respective sugars.
- Critical Step for PTS: The PTS involves multi-protein complexes. For example, the
Metabolic Network Reconstruction and Curation
- Build a Draft Model: Use an automated pipeline like the Model SEED to construct an initial draft model from your annotations [28] [26].
- Manually Curate the PTS Components:
- Add Missing PTS Reactions: Automated drafts may have gaps. Verify the presence and completeness of reactions for all relevant PTS carbohydrates (see Table 1).
- Check Reaction Stoichiometry: Ensure all PTS transport reactions correctly consume PEP and produce pyruvate.
- Balance Mass and Charge: Use tools like the COBRA Toolbox's
checkMassChargeBalancefunction to ensure all reactions are mass- and charge-balanced [29].
- Gap Filling: Run gap-filling algorithms (e.g.,
GapFindandGapFill) to identify and fill metabolic gaps that prevent the synthesis of essential biomass components [28]. This step is crucial for ensuring the model can produce PTS-related biomass precursors like peptidoglycan.
Define Constraints and Biomass Objective
- Set Exchange Reactions: Constrain the model to a specific growth medium by defining the upper and lower bounds of exchange reactions for available nutrients (e.g., glucose, oxygen).
- Define the Biomass Objective Function: The biomass equation is typically set as the objective function to be maximized during FBA [29]. Ensure this equation includes all major macromolecules, such as proteins, DNA, RNA, lipids, and cell wall components like peptidoglycan (whose synthesis depends on PTS-imported sugars like GlcNAc).
Model Simulation and Validation with FBA
- Run Flux Balance Analysis (FBA): Use a linear programming solver (e.g., GLPK, GUROBI) to maximize the biomass objective function and predict growth rates and internal flux distributions [26] [29].
- Validate the PTS Integration:
- Growth on PTS Sugars: Simulate growth on individual PTS carbohydrates (glucose, mannose, etc.) and verify that the model predicts positive growth.
- Gene Essentiality: Perform in silico gene knockout studies. Deleting general PTS genes like
ptsIorptsHshould prevent growth on all PTS sugars, while knocking out a specific permease likeptsGshould only prevent growth on glucose [29]. - Flux Analysis: Examine the flux through PTS reactions under different conditions. For example, when growing on glucose, the flux through the glucose PTS reaction should be high and coupled to glycolytic flux.
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for PTS and GSMM Research
| Reagent / Resource | Function and Application | Example / Source |
|---|---|---|
| Annotation Software | Identifies protein-encoding genes and assigns functional roles from a genome sequence. | RAST, PROKKA [26] [27] |
| Model Reconstruction Tools | Automates the construction of draft metabolic models from annotations. | Model SEED, PyFBA [28] [26] |
| Biochemistry Databases | Provides curated lists of biochemical reactions, metabolites, and EC numbers. | KEGG, MetaCyc, BiGG, Model SEED Biochemistry [28] [26] |
| Simulation & Analysis Software | Performs FBA, gap-filling, and model validation. | COBRA Toolbox, PyFBA [26] [29] |
| Linear Programming Solver | Solves the linear optimization problem at the heart of FBA. | GLPK, GUROBI, CPLEX [26] [29] |
Workflow and Pathway Visualization
The following diagram illustrates the core phosphorylation cascade of the PTS for glucose and its integration point into the metabolic network, which is crucial for understanding the stoichiometric constraints it imposes.
PTS Phosphorylation Cascade and Metabolic Integration
The overall workflow for building a GSMM and integrating the PTS, from genome to a validated model, is summarized below.
GSMM Reconstruction and PTS Integration Workflow
A Step-by-Step FBA Protocol for Modeling PTS Activity and Flux
Flux Balance Analysis (FBA) is a mathematical approach for analyzing the flow of metabolites through a metabolic network, enabling researchers to predict cellular growth rates or metabolite production. A fundamental requirement for FBA is the objective function, which defines the biological goal the cell is presumed to be optimizing. In genome-scale metabolic models, the biomass objective function (BOF) is frequently employed to simulate cellular growth. It mathematically describes the rate at which all biomass precursors—including amino acids, lipids, nucleotides, and cofactors—are synthesized in the correct proportions to support growth [30]. The selection of an appropriate objective function is particularly crucial when analyzing specific systems like the E. coli phosphotransferase system (PTS), as it directly influences the predicted flux distributions and the biological interpretation of results. The PTS is both a major carbohydrate uptake system and an important signaling unit, and accurately modeling its contribution to cellular fitness requires careful formulation of the optimization problem [31].
Theoretical Foundation: Biomass vs. ATP Maximization
The Biomass Objective Function
The biomass objective function is a stoichiometric representation of the biomass composition of a cell. Its formulation involves precise knowledge of the macromolecular makeup of the cell and the biosynthetic energy required to generate this biomass from metabolic precursors [30]. The development of a BOF can be approached at different levels of complexity:
- Basic Level: Involves defining the macromolecular content of the cell (weight fractions of protein, RNA, DNA, lipids, etc.) and the metabolic building blocks that constitute each macromolecule (e.g., amino acids for proteins). This level establishes the stoichiometric requirements for carbon, nitrogen, and other elements [30].
- Intermediate Level: Incorporates the energetic costs of polymerization. For instance, the consumption of approximately 2 ATP and 2 GTP molecules is included to account for the energy required to polymerize each amino acid into a protein. This level also considers the by-products of biosynthesis, such as water and diphosphate, which become available to the cell [30].
- Advanced Level: Expands the function to include vitamins, essential cofactors, and trace elements. An advanced approach may also involve creating a "core" biomass objective function, which defines the minimal set of components necessary for cellular viability. This core BOF can improve the accuracy of predicting gene and reaction essentiality [30].
Alternative Cellular Objectives
While biomass maximization is a common assumption, especially for microorganisms in nutrient-rich environments, studies have explored a range of potential cellular objectives. Research utilizing E. coli metabolic networks has investigated objectives such as [30]:
- Maximization of ATP yield
- Minimization of nutrient uptake
- Minimization of redox potential (minimizing NADH production)
- Minimization of total intracellular flux
No single objective function perfectly predicts metabolic behavior under all conditions. The predictive performance of an objective function can depend heavily on the environmental context, such as nutrient scarcity versus abundance [30].
The PTS as a Sensory and Metabolic Unit
The phosphotransferase system in E. coli is more than a mere transport apparatus. It functions as an integrated sensory element that measures the activity of central metabolism. The PTS, together with the phosphoenolpyruvate (PEP)/pyruvate node, constitutes a robust structure that maps the glycolytic and gluconeogenetic flux to the phosphorylation states of PTS proteins [31]. This phosphorylation state, in turn, influences downstream cellular processes, including catabolite repression and regulation of metabolic pathways. Therefore, when defining an objective function for PTS-related studies, one must consider that the PTS flux is not only a determinant of substrate uptake rate but also a reflection of the cell's physiological status [31].
Application Notes: Implementing FBA for PTS Flux Analysis
Protocol: Simulating PTS Flux with a Biomass Maximization Objective
This protocol details the steps to perform FBA on an E. coli metabolic model to analyze fluxes through the PTS, using biomass maximization as the objective function.
I. Model Preparation
- Obtain a Genome-Scale Model: Acquire a curated metabolic reconstruction of E. coli, such as the iJR904 model (1,320 reactions, 625 metabolites) or a more recent equivalent [30].
- Verify PTS Reactions: Ensure the model accurately represents the glucose PTS pathway. The key reaction is typically:
PEP + glucose_{out} -> pyruvate + glucose-6-phosphate_{in} - Set the Objective Function: Designate the biomass reaction (
BIOMASS_Ec_iJR904) as the objective function to be maximized.
II. Simulation Setup
- Define Constraints:
- Set the lower bound of the glucose exchange reaction (e.g.,
EX_glc__D_e) to a negative value (e.g., -10 mmol/gDW/h) to allow glucose uptake. - Set the lower bound of the oxygen exchange reaction (
EX_o2_e) to a negative value for aerobic conditions (e.g., -20 mmol/gDW/h) or to 0 for anaerobic conditions. - Constrain other carbon sources to zero uptake if a single carbon source is desired.
- Set the lower bound of the glucose exchange reaction (e.g.,
- Perform FBA: Solve the linear programming problem: Maximize: Z = c^T * v Subject to: S * v = 0, and lb ≤ v ≤ ub where Z is the objective function (biomass), c is a vector of zeros with a one at the position of the biomass reaction, v is the flux vector, S is the stoichiometric matrix, and lb and ub are lower and upper flux bounds [30].
III. Flux Analysis
- Inspect the Solution: The primary output is the optimal growth rate (the value of Z) and the corresponding flux distribution (v).
- Extract PTS Flux: Examine the flux value through the PTS reaction for glucose (
PTS_GLCror equivalent). Under biomass maximization with glucose as the sole carbon source, this flux is typically high. - Analyze the PEP/Pyruvate Node: Review the fluxes of reactions around PEP and pyruvate, as these are directly connected to PTS activity.
Protocol: Comparing Biomass vs. ATP Maximization Objectives
This protocol outlines a comparative analysis to evaluate how different objective functions influence the predicted PTS flux.
I. Model and Constraint Setup
- Use the same E. coli model and constraints as in Protocol 3.1.
II. Sequential FBA Simulations
- Simulation A - Biomass Maximization:
- Set the biomass reaction as the objective.
- Run FBA and record the growth rate and the flux through the PTS reaction.
- Simulation B - ATP Maximization:
- Change the objective function to maximize the flux of the ATP maintenance reaction (
ATPM). - Run FBA and record the ATP production rate and the flux through the PTS reaction.
- Change the objective function to maximize the flux of the ATP maintenance reaction (
III. Analysis and Interpretation
- Compare Flux Distributions: The PTS flux is likely to be high in both simulations, as it is an efficient route for simultaneous carbon uptake and phosphorylation. However, the overall flux distribution in central metabolism may differ significantly.
- Validate with Experimental Data: Compare the predicted flux distributions from both objectives with experimental data, such as (^{13})C-based flux measurements, if available. The objective that produces a flux distribution closer to the experimental data is more representative of the cell's physiological state under those specific conditions [30].
Table 1: Example Output from a Comparative FBA Study on E. coli Central Metabolism
| Objective Function | Predicted Growth Rate (1/h) | PTS Flux (mmol/gDW/h) | ATP Production Rate (mmol/gDW/h) | Key Characteristics |
|---|---|---|---|---|
| Maximize Biomass | 0.85 | -15.5 | 12.8 | Represents balanced, growth-oriented metabolism. |
| Maximize ATP | N/A | -18.2 | 25.5 | Often leads to unrealistic, hyper-metabolic states. |
| Minimize Redox Potential | 0.42 | -8.1 | 9.1 | May be relevant under specific stress conditions [30]. |
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Computational Tools for FBA of the E. coli PTS
| Item | Function/Description | Example/Reference |
|---|---|---|
| Genome-Scale Model | A stoichiometric reconstruction of E. coli metabolism; the core platform for FBA. | iJR904, iML1515 [30] |
| FBA Software | Computational environment for setting up and solving constraint-based models. | CobraPy, CellNetAnalyzer, OptFlux |
| PTS Mutant Strains | Genetically modified E. coli with deleted or altered PTS genes; crucial for model validation. | Strains with deletions in ptsG, ptsI, crr [32] |
| 13C-Labeled Substrates | Tracers for experimental flux determination (e.g., via 13C-MFA) to validate FBA predictions. | [1-13C]-glucose, [U-13C]-glucose |
| Biomass Composition Data | Quantitative measurements of cellular macromolecules (proteins, RNA, DNA, lipids) used to formulate the biomass objective function. | E. coli biomass spreadsheet [30] [33] |
Visualization of Metabolic Pathways and Workflows
The following diagrams illustrate the core concepts and workflows discussed in this application note.
Diagram 1: E. coli Glucose PTS Pathway. The pathway shows phosphoryl transfer from PEP to incoming glucose via PTS proteins EI and EIIA^Glc. The PEP/pyruvate ratio is a key sensory input [31].
Diagram 2: FBA Workflow for PTS Analysis. The flowchart outlines the key steps in performing FBA to analyze fluxes in the PTS, highlighting the critical decision point of selecting the objective function.
Condition-Specific Objective Functions
It is imperative to recognize that the predictive power of an objective function is context-dependent. Studies have demonstrated that while biomass maximization effectively predicts fluxes in batch cultures with excess carbon and energy, other objectives may be more accurate under different conditions. For example, in carbon-limited chemostat cultures, objectives like maximization of ATP yield or even minimization of total flux (representing a principle of metabolic economy) have shown higher correlation with experimental flux data [30]. Furthermore, the PTS itself can be subject to complex regulation, such as cross-talk with other PTS branches (e.g., the PTSNtr), which can influence the relationship between PTS flux and global cellular objectives [31].
Defining the objective function is a critical step in employing FBA to investigate the E. coli PTS. The biomass objective function provides a robust and biologically relevant objective for simulating growth under many standard conditions and is an excellent starting point for analysis. However, researchers should be aware that ATP maximization and other objectives can serve as valuable comparative tools, helping to explore alternative metabolic states and to interpret flux distributions when biomass maximization fails to match experimental data. The optimal choice of objective function ultimately depends on the specific physiological context of the cells being modeled. A rigorous, iterative process of model prediction followed by experimental validation remains the best practice for establishing a reliable FBA framework for PTS research.
Flux Balance Analysis (FBA) serves as a cornerstone computational technique for predicting metabolic phenotypes in Escherichia coli and other organisms. This method operates on stoichiometric genome-scale metabolic models and employs linear programming to predict steady-state metabolic fluxes, typically optimizing for objectives such as biomass production. The accuracy and predictive power of FBA are profoundly enhanced when the solution space is constrained using experimentally measured uptake and secretion rates. This document provides detailed application notes and protocols for the systematic integration of such experimental data, with a specific focus on research involving the Phosphotransferase System (PTS) in E. coli. The PTS is a major carbohydrate uptake system in bacteria, and its accurate representation in metabolic models is crucial for simulating carbon metabolism accurately [15] [34].
Quantitative Data Compilation for PTS and Central Metabolism
The first critical step in model constraint is the compilation of experimentally determined flux data. The table below summarizes exemplary quantitative data for key transport and metabolic fluxes relevant to PTS substrates and central metabolism in E. coli. These values, often reported as mmol/gDW/h, can be directly used as constraints in FBA simulations.
Table 1: Experimentally Measured Flux Data for Model Constraint
| Reaction or Metabolite | Experimental Flux Value (mmol/gDW/h) | Condition / Strain | Notes |
|---|---|---|---|
| Glucose uptake (via PTS) | -8.2 to -12.5 [35] | Wild-type E. coli (various models) | Aerobic, glucose minimal medium. Negative sign denotes uptake. |
| Sucrose uptake (SUCpts) | Constrain upper bound | Model iJR904 [36] | Reaction: pep_c + sucr_e -> pyr_c + suc6p_c |
| D-sorbitol uptake (SBTpts) | Constrain upper bound | Universal BiGG Reaction [37] | Reaction: pep_c + sbt__D_e ⇌ pyr_c + sbt6p_c |
| Acetate secretion | +2.1 to +5.8 [35] | Wild-type and mutant strains | Secretion flux is positive. |
| Biomass yield | ~0.45 (g biomass / g glucose) [35] | Optimal growth prediction | Highly sensitive to model constraints [38]. |
| Pyruvate secretion | Variable | e.g., Δpgi mutant [35] | Can be significantly elevated in knockout strains. |
Detailed Protocol for Data Integration and Model Constraint
This protocol outlines the procedure for integrating experimentally measured uptake and secretion rates into an FBA model, using tools such as the COBRA Toolbox or PyFBA.
Prerequisites and Software Requirements
- A curated genome-scale metabolic model of E. coli (e.g., iJR904, iAF1260, or k-ecoli457) [26] [35].
- Experimentally determined uptake and secretion rates for the strain and condition of interest, as shown in Table 1.
- Computational software: The PyFBA package in Python or the COBRA Toolbox in MATLAB. PyFBA is an open-source Python-based software package that facilitates the reconstruction of metabolic models from functional annotations and allows running FBA under different constraints [26].
Step-by-Step Workflow
Step 1: Model Import and Validation Import the metabolic model into your chosen software environment. Validate the model by checking for mass and charge balance in key reactions, especially those related to PTS transport (e.g., glucose, sucrose uptake). Ensure the model can produce a feasible solution for growth on a minimal medium before applying new constraints.
Step 2: Mapping Experimental Data to Model Reactions
Map each measured extracellular metabolite to its corresponding exchange reaction in the model. For example, map the measured glucose uptake rate to the reaction EX_glc__D_e or the specific PTS reaction GLCpts. For PTS sugars, note that the uptake reaction involves phosphoenolpyruvate (PEP) and produces pyruvate and the phosphorylated sugar internally (e.g., pep_c + glc__D_e -> pyr_c + g6p_c) [36] [37] [39].
Step 3: Setting Flux Bounds
Apply the measured flux values as constraints by setting the lower and upper bounds (lb, ub) of the corresponding exchange reactions.
- For an uptake rate of
-10 mmol/gDW/h, set the lower bound (lb) of the exchange reaction to-10and the upper bound (ub) to0or a small positive value (e.g.,999999) if secretion is possible. - For a secretion rate of
+3.5 mmol/gDW/h, set the lower bound to-0(or a large negative value for uptake) and the upper bound to+3.5.
Step 4: Implementing Complex PTS Constraints When constraining a PTS reaction directly, remember it is a bidirectional reaction that consumes PEP and produces pyruvate. Constraining its flux will inherently affect the central energy metabolism ratios. Ensure the model's objective function (e.g., biomass production) is set.
Step 5: Model Simulation and Gap Analysis Run FBA to obtain a flux distribution. If the model fails to grow or shows unrealistic fluxes, perform a gap analysis. This involves checking for dead-end metabolites and blocked reactions, particularly around the transport and initial metabolism of the constrained carbon source. Gap-filling protocols, as implemented in PyFBA and Model SEED, can be used to add missing reactions necessary to support growth, provided there is genomic or biochemical evidence [26].
Step 6: Validation and Iteration Validate the constrained model by comparing its predictions against other experimental data not used for constraining, such as the secretion rates of other by-products (e.g., acetate, formate) or growth rates. Iterate the process by adjusting constraints or investigating model topology if predictions are inconsistent with experimental observations.
Workflow Visualization
The diagram below illustrates the logical workflow for constraining a metabolic model with experimental data.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Research Reagents and Resources for PTS and FBA Research
| Reagent / Resource | Function / Description | Example / Source |
|---|---|---|
| Genome-Scale Metabolic Model | A stoichiometric matrix representing all known metabolic reactions in E. coli; the core structure for FBA. | iJR904, iAF1260, k-ecoli457 [40] [35] |
| Software for FBA | Computational platform to build models, apply constraints, and solve the linear programming problem. | PyFBA [26], COBRA Toolbox |
| Annotation Pipeline | Tool to identify protein-encoding genes and assign functional roles from raw genome sequence. | RAST, PROKKA [26] |
| Biochemistry Database | A curated database linking functional roles to enzyme complexes and biochemical reactions. | Model SEED [26] |
| Linear Programming Solver | The computational engine that performs the optimization to find a flux distribution. | GLPK, IBM ILOG CPLEX [26] |
| Lipid Nanodiscs | A native-like lipid environment for reconstituting membrane proteins for structural studies. | Used in Cryo-EM of IICBGlc [15] |
| n-Dodecyl-β-D-Maltopyranoside (DDM) | A detergent used to solubilize and purify membrane proteins for structural biology. | Used to stall IICBGlc in intermediate state [34] |
Escherichia coli possesses a remarkable capacity to utilize diverse carbon sources, with a fundamental dichotomy existing between sugars imported via the phosphoenolpyruvate-dependent phosphotransferase system (PTS) and those transported by non-PTS mechanisms such as major facilitator superfamily (MFS) proton symporters or ATP-binding cassette (ABC) transporters [41]. The PTS is a group translocation system that couples sugar transport with phosphorylation, using phosphoenolpyruvate (PEP) as the phosphoryl donor and energy source [42] [15]. This system is of paramount interest in metabolic modeling because it directly links substrate uptake to central carbon metabolism and exerts profound regulatory control over cellular physiology. Understanding the distinct metabolic and regulatory consequences of growth on PTS versus non-PTS sugars is essential for accurate simulation of E. coli metabolism, particularly in the context of flux balance analysis (FBA) which relies on stoichiometric representation of these biochemical transformations [43].
The genome of E. coli encodes numerous transport systems, with 97 proteins identified as involved in sugar transport [41]. However, the preferential use of specific systems, particularly the glucose-specific PTS transporter IICBGlc (encoded by ptsG), creates a hierarchical structure of carbon source utilization that must be captured in metabolic models [44] [45]. This application note provides a structured framework for simulating growth on different carbon sources, with specific protocols for model formulation, data integration, and computational analysis tailored to PTS and non-PTS substrates.
Theoretical Background and Key Physiological Differences
The Phosphotransferase System: Mechanism and Regulation
The PTS represents a sophisticated biochemical pathway where energy transfer and solute transport are mechanistically coupled. The system consists of cytoplasmic and membrane-associated components that form a phosphorylation cascade: Enzyme I (EI, encoded by ptsI) and the histidine-containing phosphocarrier protein (HPr, encoded by ptsH) are general proteins common to all PTS sugars, while Enzyme II (EII) complexes are sugar-specific [42] [15]. For glucose transport, the EII complex comprises IIAGlc (encoded by crr) and the membrane transporter IICBGlc (encoded by ptsG).
The phosphorylation cascade begins with PEP phosphorylating EI, which subsequently transfers the phosphoryl group to HPr. Phospho-HPr then donates the phosphoryl group to the sugar-specific IIA domain, which in turn phosphorylates the IIB domain. Finally, the IIB domain phosphorylates the sugar molecule as it is transported across the membrane by the IIC domain [42] [15]. This intricate process results in the simultaneous transport and phosphorylation of the sugar substrate at the expense of PEP.
Recent structural insights from cryo-EM studies have elucidated the molecular mechanism of the glucose-specific IICBGlc transporter, revealing an elevator-type transport mechanism where the transport domain undergoes a substantial vertical shift of approximately 13 Å, presenting the substrate binding site to either the cytoplasmic or periplasmic side of the membrane [15]. This structural information provides a basis for understanding the kinetics and specificity of PTS transport.
Table 1: Key Components of the E. coli PTS System for Glucose Transport
| Component | Gene | Function | Location |
|---|---|---|---|
| Enzyme I | ptsI | Autophosphorylation using PEP | Cytoplasm |
| HPr | ptsH | Phosphocarrier between EI and EII | Cytoplasm |
| IIAGlc | crr | Sugar-specific phosphotransfer | Cytoplasm/Membrane |
| IICBGlc | ptsG | Glucose transport and phosphorylation | Membrane |
Regulatory Networks Governing Carbon Source Preference
The PTS serves as a central regulatory node that monitors carbon availability and coordinates cellular metabolism accordingly. The phosphorylation state of IIAGlc acts as a key metabolic signal that influences multiple cellular processes:
Inducer Exclusion: Dephosphorylated IIAGlc binds to and inhibits several non-PTS uptake systems, preventing entry of their respective inducers [42] [46]. This mechanism ensures preferential utilization of PTS sugars over non-PTS alternatives.
cAMP-CRP Regulation: The phosphorylation state of IIAGlc influences adenylate cyclase activity, thereby modulating intracellular cAMP levels [20] [44]. cAMP complexes with CRP (cAMP receptor protein) to activate transcription of numerous catabolic operons, including those for non-PTS sugars.
Mlc Regulation: The global repressor Mlc controls expression of ptsG and other glucose-related genes. During glucose transport, dephosphorylated IICBGlc sequesters Mlc to the membrane, derepressing its target genes [45].
These interconnected regulatory mechanisms create a sophisticated hierarchy of carbon source utilization. Experimental studies have established that E. coli exhibits a defined "soft" hierarchy for non-PTS sugars, where the sugar supporting faster growth rate dominates in mixtures [44]. The hierarchy among common non-PTS sugars is: lactose > arabinose > xylose > sorbitol > rhamnose > ribose, which corresponds directly to the growth rate supported by each sugar as a sole carbon source [44].
Growth Parameters and Metabolic Flux Distributions
Different carbon sources support distinct growth rates and metabolic flux distributions in E. coli due to their different entry points into central metabolism and varying ATP yields per mole of carbon. PTS sugars typically support faster growth rates than non-PTS sugars because of their efficient transport and immediate phosphorylation without ATP expenditure.
Table 2: Experimentally Determined Growth Rates on Different Carbon Sources
| Carbon Source | Transport System | Maximal Growth Rate (h⁻¹) | Entry Point to Metabolism |
|---|---|---|---|
| Glucose | PTS | 0.53-0.60 [44] | Glucose-6-phosphate |
| Lactose | MFS (LacY) | 0.53 ± 0.01 [44] | Glucose (after hydrolysis) |
| Arabinose | ABC/MFS | 0.47 ± 0.01 [44] | D-Xylulose-5-phosphate |
| Xylose | ABC/MFS | 0.43 ± 0.01 [44] | D-Xylulose-5-phosphate |
| Sorbitol | PTS | 0.37 ± 0.01 [44] | Fructose-6-phosphate |
| Rhamnose | PTS? | 0.32 ± 0.01 [44] | Dihydroxyacetone-P + L-Lactaldehyde |
| Ribose | ABC | 0.29 ± 0.01 [44] | D-Ribose-5-phosphate |
The entry point of carbon into metabolic networks fundamentally influences the optimal utilization strategy. Carbon sources entering the upper glycolysis (Group A) typically exhibit diauxie when mixed with other Group A sources, while mixtures of Group A and Group B sources (entering at other points) often show co-utilization [47]. This behavior emerges from optimal protein resource allocation constrained by network topology [47].
Metabolic Modeling Considerations for PTS vs. Non-PTS Transport
When constructing FBA models for simulating growth on different carbon sources, key stoichiometric distinctions must be captured:
PTS Transport: Consumes 1 PEP and produces 1 pyruvate per glucose molecule transported [42]:
Glucose_external + PEP → Glucose-6-phosphate + PyruvateNon-PTS Transport: Typically consumes 1 ATP per glucose (ABC transporters) or uses proton symport without direct ATP consumption (MFS transporters), though the latter still incurs energetic costs for maintaining proton gradients [41].
Energy Stoichiometry: The PTS "saves" one ATP equivalent compared to non-PTS transport followed by hexokinase phosphorylation (which consumes ATP), creating a fundamental energetic distinction that influences predictions of growth yield and metabolic flux.
These stoichiometric differences significantly impact FBA simulations, particularly when predicting carbon source preferences or engineering transport systems for biotechnological applications.
Protocol for FBA of Carbon Source Utilization
Model Reconstruction and Curation
Materials:
- Genome-scale metabolic model of E. coli (e.g., iJO1366, EcoCyc)
- Annotation databases (KEGG, BioCyc, UniProt)
- Computational tools for gap-filling (e.g., ModelSEED, CarveMe)
- Stoichiometric database for transport reactions
Procedure:
Identify Transport Systems: For each carbon source of interest, identify the specific transport system(s) using genomic annotations and biochemical databases [41]. E. coli K-12 MG1655 possesses 97 proteins involved in sugar transport, with multiple systems potentially available for a single sugar.
Formulate Stoichiometric Equations: Precisely define the stoichiometry for each transport reaction:
- For PTS sugars: Include PEP consumption and pyruvate production
- For ABC transporters: Include ATP hydrolysis
- For MFS symporters: Include proton stoichiometry
Add Regulatory Constraints: Implement constraints that reflect known regulatory phenomena:
- Add capacity constraints for inducer exclusion (limiting simultaneous uptake of certain sugars)
- Incorporate hierarchical utilization patterns based on experimental data [44]
Validate with Experimental Data: Compare simulated growth rates and substrate uptake rates with published experimental values (see Table 2) and iteratively refine the model.
Materials:
- Constrained-based modeling software (CobraPy, COBRA Toolbox)
- Experimental growth data for validation
- Optimization solver (Gurobi, CPLEX)
Procedure:
Define Simulation Conditions: Specify the medium composition with precise concentrations of each carbon source.
Implement Dynamic Constraints: For sequential utilization (diauxie), implement dynamic constraints that:
- Switch off uptake of secondary carbon sources when the preferred source is available
- Adjust uptake rates based on the phosphorylation state of IIAGlc [46]
Run FBA Simulations: Maximize biomass production subject to the defined constraints to predict:
- Growth rates
- Substrate uptake rates
- Metabolic flux distributions
- Potential byproduct secretion
Validate with Experimental Hierarchy: Compare predicted carbon source preference with the established hierarchy [44] and adjust regulatory constraints if necessary.
Research Reagent Solutions
Table 3: Essential Research Reagents for Studying PTS and Non-PTS Sugar Transport
| Reagent/Category | Specific Examples | Function/Application | Experimental Use |
|---|---|---|---|
| PTS Components | Enzyme I, HPr, IIAGlc, IICBGlc | Study phosphotransfer mechanism | In vitro phosphorylation assays; Transport studies |
| Non-PTS Transporters | LacY (lactose), AraE (arabinose), XylE (xylose) | Comparative transport studies | Membrane vesicle transport assays |
| Metabolic Intermediates | PEP, Pyruvate, Glucose-6-P | Quantify metabolic fluxes | Metabolite profiling; ¹³C flux analysis |
| Reporter Systems | GFP transcriptional fusions (lacZYA, araBAD, xylAB) | Monitor promoter activity | Real-time gene expression in sugar mixtures |
| Inhibitors/Modulators | α-Methylglucoside (non-metabolizable PTS substrate) | Probe PTS function without metabolism | Induction studies; Competition experiments |
Signaling Pathway Diagrams
PTS Transport and Regulatory Network
Diagram 1: PTS Transport and Regulatory Network. The diagram illustrates the phosphorylation cascade from PEP to glucose transport, and the key regulatory outputs controlling carbon metabolism.
Carbon Source Hierarchy and Decision Logic
Diagram 2: Carbon Source Hierarchy and Decision Logic. The diagram illustrates the cellular decision-making process when multiple carbon sources are available, leading to either sequential (diauxic) or simultaneous (co-utilization) consumption patterns.
The structured simulation of E. coli growth on PTS versus non-PTS sugars requires careful consideration of both stoichiometric and regulatory factors. The protocols outlined in this application note provide a systematic approach for incorporating these factors into FBA simulations, enabling more accurate predictions of metabolic behavior in complex nutrient environments. The key distinction in energy stoichiometry between PTS and non-PTS transport systems fundamentally influences cellular energetics and must be properly represented in metabolic models.
Understanding these principles has significant applications in metabolic engineering and biotechnology. For instance, engineering co-utilization of mixed sugars from lignocellulosic hydrolysates (containing both hexoses and pentoses) requires overcoming the native hierarchical regulation [44] [47]. Similarly, optimizing product yields in industrial bioprocesses depends on accurate prediction of carbon allocation under different nutrient conditions. The integration of quantitative physiological data with genome-scale models, as demonstrated herein, provides a powerful framework for both basic research and applied biotechnology.
Flux balance analysis (FBA) of the bacterial phosphotransolpyruvate-dependent phosphotransferase system (PTS) provides critical insights into cellular energy management and carbon source utilization. The PTS represents a fundamental link between signal transduction and cellular physiology, acting as a sensory element that measures activity of central metabolism [31]. In Escherichia coli, the glucose-specific transporter IICBGlc serves as the major glucose transporter and functions as an essential PTS component, coupling vectorial transmembrane transport with concomitant phosphorylation of carbohydrates [15]. This system functions not merely as a transport mechanism but as a sophisticated regulatory network that influences gene expression, metabolic regulation, and ultimately, bacterial competitiveness in changing environments.
Understanding flux distributions through the PTS requires integration of structural biology, kinetic parameters, and systems biology approaches. Recent cryo-electron microscopy (cryo-EM) structures of the glucose-bound IICBGlc protein have revealed the molecular architecture of this transporter in lipid nanodiscs, capturing both occluded inward-facing and outward-facing conformations [15]. These structural insights, combined with molecular dynamics simulations, provide the physical basis for interpreting phosphate transfer kinetics and sugar internalization rates essential for accurate FBA modeling of PTS function in E. coli.
Quantitative Parameters for PTS Flux Modeling
Kinetic and Thermodynamic Constants
Table 1: Experimentally Determined Kinetic Parameters for E. coli PTS Components
| Parameter | Value | Experimental Context | Significance in Flux Modeling |
|---|---|---|---|
| Glucose uptake rate (P. putida) | 1.74 ± 0.29 g/g/h (glucose), 0.31 ± 0.02 g/g/h (xylose) | Sequential consumption in wild-type strain [48] | Establishes baseline transport kinetics for model validation |
| Glucose uptake rate (mutant) | 0.23 ± 0.04 g/g/h (glucose), 0.19 ± 0.01 g/g/h (xylose) | Co-consumption in hexokinase mutant [48] | Demonstrates flux redistribution when phosphorylation capacity is reduced |
| PTS protein phosphorylation equilibrium constants | Variable based on PEP/pyruvate ratio | Mathematical modeling of P. putida PTS [31] | Determines phosphate flow distribution between PTS branches |
| Vertical TD domain shift | ~13 Å | Structural analysis of IICBGlc conformations [15] | Informs transport rate calculations based on conformational changes |
Table 2: Structural and Biophysical Parameters for IICBGlc Transporter
| Parameter | Inward-Facing State | Outward-Facing State | Method of Determination |
|---|---|---|---|
| RMSD (TD domains) | 0.83 Å | 0.83 Å | Cryo-EM structure alignment [15] |
| RMSD (SD domains) | 1.05 Å | 1.05 Å | Cryo-EM structure alignment [15] |
| TM5-AH angle | Reference | +23° | Molecular dynamics simulation [15] |
| Dimer interface area | ~2800 Ų | ~2800 Ų | Cryo-EM structural analysis [15] |
| Key hinge residues | P173/P174 double proline motif | P173/P174 double proline motif | Structural analysis [15] |
Experimental Protocols for PTS Flux Analysis
Protocol 1: In Vivo Phosphorylation State Analysis
Purpose: To determine the phosphorylation state of PTS proteins under different metabolic conditions for flux distribution calculations.
Materials:
- PTS-specific antibodies (e.g., affinity-purified anti-C terminal antibody)
- Dielectric breakdown apparatus for cell lysis
- Western blotting equipment and enhanced chemiluminescent substrate
- [32P]orthophosphate (0.18 Ci/μmol)
- Ice-cold dilution and wash buffers (Tris-succinate)
- Filtration system (Whatman GF/F filters)
- Liquid scintillation spectrometry equipment
Procedure:
- Culture E. coli cells under targeted conditions (varying carbon sources, growth phases)
- Withdraw samples aseptically at specified time points during growth phase transitions
- Centrifuge at 2,300 × g for 10 minutes and wash with appropriate buffer
- For radioactive tracing: Incubate cell suspension with [32P]orthophosphate for 1 minute at 25°C
- Terminate phosphorylation by adding ice-cold dilution buffer
- Immediately filter samples and wash filters with cold solution
- Determine radioactivity retained on filters using liquid scintillation spectrometry
- For Western blot: Prepare membrane fraction, separate proteins using SDS-PAGE (12% polyacrylamide)
- Transfer to PVDF membranes and detect using PTS-specific antibodies
- Quantify phosphorylation states using densitometry analysis
Data Interpretation: The phosphorylation state reflects the flux distribution through PTS branches. Higher phosphorylation indicates greater phosphate flow through that pathway [31].
Protocol 2: Structural Analysis of PTS Transporters in Lipid Nanodiscs
Purpose: To obtain high-resolution structures of PTS transporters in native-like lipid environments for mechanistic insights into transport rates.
Materials:
- Purified, monodisperse full-length IICBGlc protein
- Lipid nanodisc components (membrane scaffold proteins, lipids)
- Cryo-EM equipment (electron microscope, cryo-holder)
- CryoSPARC or similar single-particle analysis software
- Molecular dynamics simulation software
Procedure:
- Reconstitute pure IICBGlc protein in lipid nanodiscs to create native-like lipid environment
- Add glucose substrate to protein preparation
- Perform cryo-EM image acquisition with appropriate parameters
- Process images using cryoSPARC, performing 2D classification
- Refine distinct particle populations to 3D density maps
- Build atomic models into density maps representing different conformational states
- Perform molecular dynamics simulations to analyze transport mechanics
- Measure structural parameters (RMSD, domain shifts, interface areas)
- Identify gating mechanisms and substrate recognition elements
Application to Flux Analysis: Structural data informs kinetic models by revealing conformational changes and gating mechanisms that limit transport rates [15].
Protocol 3: Metabolic Flux Balance Analysis of PTS
Purpose: To determine flux distributions in PTS networks using constraint-based modeling.
Materials:
- Stoichiometric model of E. coli central metabolism including PTS reactions
- Experimentally determined uptake and excretion rates
- FBA software (e.g., COBRA toolbox, specific in-house implementations)
- Physiological data (growth rates, substrate consumption)
Procedure:
- Construct stoichiometric matrix incorporating all PTS reactions
- Incorporate constraints from experimental measurements (e.g., maximum glucose uptake rate)
- Define objective function (e.g., biomass maximization)
- Solve linear programming problem to obtain flux distribution
- Validate model predictions with experimental phosphorylation states
- Perform sensitivity analysis on key parameters (PEP/pyruvate ratio, kinase activities)
- Calculate flux control coefficients for PTS enzymes
Integration with PTS Modeling: FBA provides quantitative flux distributions that can be correlated with phosphorylation states of PTS proteins to understand system regulation [31].
Research Reagent Solutions
Table 3: Essential Research Reagents for PTS Flux Analysis
| Reagent/Category | Specific Examples | Function in PTS Research |
|---|---|---|
| Radioactive Tracers | [32P]orthophosphate (0.18 Ci/μmol) [49] | Labels phosphate groups to track transfer through PTS cascade |
| Antibodies | Affinity-purified Pho84 anti-C terminal antibody [49] | Detects specific PTS proteins and phosphorylation states in Western blot |
| Structural Biology Tools | Lipid nanodiscs [15] | Provides native-like membrane environment for structural studies of transporters |
| Inhibitors/Mutants | Hexokinase mutants (Hxk1, Hxk2, Glk1) [48] | Reduces glucose phosphorylation rate to study flux redistribution |
| Mathematical Modeling Platforms | Custom MATLAB/Python implementations | Performs FBA and kinetic modeling of PTS flux distributions [31] |
Signaling Pathway and Metabolic Integration Visualizations
PTS Phosphotransfer Cascade and Metabolic Integration
PTS Flux Analysis Experimental Workflow
Interpretation Guidelines for Flux Distribution Data
Correlating Phosphorylation States with Metabolic Conditions
The phosphorylation state of PTS proteins serves as a quantitative indicator of flux distribution. Studies in Pseudomonas putida demonstrate that the phosphorylation state of PtsN, the final protein in the PTSNtr branch, varies significantly with growth phase and carbon source [31]. During rapid growth, non-phosphorylated PtsN predominates, while phosphorylated PtsN accumulates in stationary phase. This pattern reflects the reallocation of phosphate flux in response to metabolic demands. When interpreting phosphorylation data, researchers should note:
- Carbon source effects: Phosphorylation patterns differ significantly between glycolytic and gluconeogenic carbon sources
- Growth phase dependence: Late exponential phase typically shows maximal phosphorylation of terminal PTS components
- Cross-talk indicators: Phosphorylation of PTSNtr components by sugar-specific PTS branches indicates active communication between systems
Integrating Structural and Kinetic Data for Transport Rate Calculations
The elevator-type transport mechanism identified in IICBGlc structures provides a physical basis for calculating sugar internalization rates [15]. Key structural parameters for flux calculations include:
- Domain displacement: The ~13 Å vertical shift of the transport domain between inward-facing and outward-facing states correlates with substrate translocation capacity
- Gating kinetics: The identified gating residues control substrate accessibility and can be incorporated into Michaelis-Menten approximations of transport rates
- Dimer interface stability: The extensive 2800 Ų dimerization interface suggests cooperative effects that may influence transport kinetics
FBA Model Validation Using PTS Parameters
Flux balance analysis models of PTS function should be validated against experimental measurements:
- PEP/pyruvate ratio correlation: The phosphorylation state of PTS proteins should correlate with the PEP/pyruvate ratio, reflecting the energy charge available for phosphorylation
- Cross-talk quantification: In systems with multiple PTS branches (e.g., PTSFru and PTSNtr), FBA should accurately predict phosphate exchange between branches under different nutrient conditions
- Mutant validation: Models should correctly predict flux redistribution in kinase/transporter mutants, such as the reduced glucose consumption but maintained xylose consumption in hexokinase mutants [48]
These application notes provide a comprehensive framework for analyzing flux distributions in the E. coli PTS, integrating structural biology, kinetic measurements, and systems biology approaches to enable accurate interpretation of phosphate transfer and sugar internalization rates in the context of drug development and metabolic engineering research.
The Phosphotransferase System (PTS) is a major bacterial carbohydrate uptake system that uses phosphoenolpyruvate (PEP) as an energy source to phosphorylate and import sugars across the cytoplasmic membrane [14]. In Escherichia coli, the fructose-specific PTS (PTSFru) is a well-characterized uptake route. However, a complete understanding of fructose metabolism requires analyzing its interaction with a second, non-sugar-transporting PTS branch, the PTSNtr (Nitrogen-related PTS), which is involved in signal transduction and metabolic regulation [31]. This case study demonstrates the application of a Flux Balance Analysis (FBA) protocol to analyze the interaction between PTSFru and PTSNtr, providing a framework for researchers to simulate and predict metabolic outcomes under varying genetic and environmental conditions.
Quantitative Data on Fructose Uptake and PTS Systems
Proteomic Upregulation of PTS Components on Fructose
Quantitative proteomic data from related bacterial systems highlight the significant metabolic investment required for fructose utilization. The following table summarizes the upregulation of PTSFru components in Azospirillum brasilense when fructose is the sole carbon source, compared to malate [50].
Table 1: Upregulation of Fructose PTS (PTSFru) Components
| Protein | Gene Symbol | Function | Abundance Ratio (Fructose/Malate) |
|---|---|---|---|
| FruB | fruB |
EIIA component of PTS | 44.51 |
| FruK | fruK |
1-phosphofructokinase | 12.01 |
| FruA | fruA |
EIIBC membrane component | 3.84 |
Flux Distribution Between PTS Branches
Mathematical modeling of Pseudomonas putida, which possesses analogous PTS branches, reveals a key functional interaction. During growth on fructose, a significant portion of the phosphoryl flux through the PTS system is provided by the PTSNtr branch, demonstrating a critical cross-talk between the two systems [31].
Table 2: Flux Distribution in PTS Branches During Growth on Fructose
| PTS Branch | Primary Function | Key Proteins | Proposed Role During Fructose Uptake |
|---|---|---|---|
| PTSFru (C-branch) | Fructose uptake & phosphorylation | FruA, FruB, FruK | Direct phosphorylation and import of fructose |
| PTSNtr | Signal transduction & metabolic regulation | PtsP, PtO, PtsN | Major provider of phosphoryl groups during fructose uptake |
Experimental Protocols for Key Analyses
Protocol 1: In Silico Flux Balance Analysis (FBA) of PTS Interactions
Purpose: To predict the growth rate and internal flux distribution of E. coli under different conditions, and to quantify the metabolic interaction between PTSFru and PTSNtr.
Materials:
- A curated Genome-Scale Metabolic Model (GEM) of E. coli (e.g., iJO1366).
- FBA software (e.g., COBRA Toolbox for MATLAB/Python, or the specific tools mentioned in [51]).
- A defined minimal medium composition for simulations.
Methodology:
- Model Curation: Verify that the GEM accurately represents both the PTSFru (involving
fruB,fruA,fruK) and PTSNtr (involvingptsP,ptsO,ptsN) systems. - Define Constraints:
- Set the fructose uptake rate as the sole carbon source (e.g., to a physiologically relevant value like 10 mmol/gDW/h).
- Constrain the uptake rates for all other carbon sources to zero.
- Set constraints for oxygen and other essential nutrients.
- Simulate Monoculture Growth: Perform FBA with the objective of maximizing biomass growth. Record the optimal growth rate and the flux through key reactions, particularly:
- The fructose uptake reaction (PTSFru).
- The phosphate transfer reactions within PTSNtr (PtsP/PtsO/PtsN).
- Analyze Cross-Talk: To investigate the phosphoryl flux from PTSNtr to PTSFru, create a simulation where the direct phosphorylation flux from PEP to PTSFru is constrained to zero. The resulting growth rate (if any) and the required flux through PTSNtr will indicate the capacity and necessity of cross-talk.
- Validation with Mutant Data: Simulate knockout mutants (e.g.,
ΔptsPorΔfruB) and compare the predicted growth phenotypes on fructose with experimental data from the literature to validate the model.
Protocol 2: Assessing Fructose-Dependent Regulation of Associated Systems
Purpose: To experimentally determine the effect of fructose availability on the expression of systems like the Type VI Secretion System (T6SS), which may be co-regulated with fructose uptake.
Materials:
- Wild-type A. brasilense Sp7 (or a suitable E. coli model strain).
- An isogenic mutant with a disrupted T6SS core component.
- Minimal media with either 40 mM malate or 40 mM fructose as the sole carbon source [50].
Methodology:
- Culture Conditions: Grow biological triplicates of the wild-type and mutant strains in minimal media with either fructose or malate as the carbon source.
- Proteomic Analysis:
- Harvest cells at the mid-exponential growth phase (OD600 ≈ 1.0).
- Extract proteins and perform quantitative proteomics (e.g., LC-MS/MS).
- Identify proteins significantly upregulated in the fructose condition compared to the malate condition.
- Co-culture Inhibition Assay:
- To test T6SS functionality, co-culture the wild-type strain with a target bacterium (e.g., E. coli or Agrobacterium tumefaciens) on fructose-containing solid media.
- In parallel, perform the same assay with the T6SS mutant.
- After incubation, observe the zone of growth inhibition around the test strain [50].
- Data Interpretation: Correlate the upregulation of T6SS proteins from the proteomic data with the contact-dependent growth inhibition phenotype observed in the co-culture assay.
Pathway Diagrams and Workflows
Phosphate Transfer and Cross-Talk Between PTSFru and PTSNtr
This diagram illustrates the flow of phosphoryl groups from PEP through the two PTS branches and highlights the potential cross-talk during fructose metabolism, as suggested by studies in P. putida [31].
FBA Workflow for Analyzing PTS Interactions
This workflow outlines the computational protocol for applying FBA to study the metabolic interplay between the PTSFru and PTSNtr systems.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Tools for PTS-FBA Research
| Item | Function/Description | Example Use Case |
|---|---|---|
| Curated E. coli GEM | A genome-scale metabolic model serving as the in silico representation of metabolism. | Foundation for all FBA simulations; e.g., iJO1366. |
| FBA Software Platform | Computational tools for constraint-based modeling (e.g., COBRA Toolbox, COMETS, MICOM). | Performing flux balance analysis and community modeling [51]. |
| Defined Minimal Media | Chemically defined media with specific carbon sources to control nutrient input. | Culturing cells for proteomics or validating FBA predictions [50]. |
| PTS Gene Knockout Strains | Isogenic mutant strains with deletions in specific PTS genes (e.g., ΔfruB, ΔptsP). | Validating model predictions and studying the functional role of individual PTS components. |
| Quantitative Proteomics | LC-MS/MS methods for measuring protein abundance changes. | Identifying fructose-inducible proteins and regulatory networks [50]. |
Solving Common FBA-PTS Model Issues and Optimizing Predictive Power
Flux Balance Analysis (FBA) has become an indispensable tool for predicting microbial growth and metabolic capabilities. However, significant prediction errors often occur when modeling complex regulatory networks, particularly the Phosphoenolpyruvate-Carbohydrate Phosphotransferase System (PTS) in Escherichia coli. The PTS serves as the primary transport system for numerous sugars in bacteria and plays a crucial regulatory role in carbon catabolite repression (CCR), significantly influencing bacterial growth rates and substrate preference [52]. Discrepancies between FBA predictions and experimental growth data frequently originate from incomplete annotation of PTS components and their regulatory interactions within central metabolic networks. This application note examines the sources of these prediction errors and provides detailed protocols for model refinement and experimental validation, framed within the context of FBA protocols for E. coli PTS research.
Background
The Phosphotransferase System (PTS): Structure and Function
The PTS is a complex transport and signaling system that mediates the uptake and phosphorylation of numerous sugars in E. coli. Its core architecture consists of a phosphorylation cascade that begins with phosphoenolpyruvate (PEP) and transfers phosphoryl groups through general cytoplasmic components (Enzyme I and HPr) to sugar-specific Enzyme II (EII) complexes [53]. Each EII complex typically contains three subunits (EIIA, EIIB, and EIIC) that may exist as separate proteins or fused domains [53].
Beyond its transport function, the PTS operates as a global sensory network that monitors sugar influx and regulates cellular physiology accordingly. Recent research demonstrates that despite its structural complexity, the core PTS network functions in a "surprisingly simple way," sensing the overall influx of PTS sugars regardless of sugar identity and distributing this information throughout its network [53]. This integrated metabolic information then influences diverse cellular processes, including chemotaxis, catabolite repression, and metabolic gene expression [54] [53].
Central Metabolism Modeling Challenges
Genome-scale metabolic models provide comprehensive representations of metabolic networks but present computational challenges for detailed analysis of central metabolism. The EColiCore2 model addresses this limitation as a carefully reduced representation of E. coli's central metabolism derived from the genome-scale model iJO1366 [55]. This reference network preserves key phenotypic capabilities while enabling more tractable computational analyses than its genome-scale parent.
A fundamental challenge in metabolic modeling arises from the winner-take-all dynamics inherent in PTS regulation. When multiple PTS sugars are present, the system exhibits diauxic growth – a discontinuous switching behavior where cells preferentially consume one sugar before transitioning to others [16]. This metabolic switching creates non-linear growth patterns that are particularly difficult to predict using standard FBA approaches without proper network annotation and constraint settings [16].
Quantitative Analysis of Prediction Errors
Essential Reactions in Central Metabolism
Robustness analyses of E. coli metabolic networks have identified critical dependencies within central metabolism that contribute to growth prediction inaccuracies. The table below summarizes seven essential reactions in central metabolic pathways whose perturbation significantly affects growth predictions:
Table 1: Essential Reactions in E. coli Central Metabolism and Their Robustness
| Reaction | Metabolic Pathway | Gene Product | Minimum Flux (% of optimal) | Impact on Growth Prediction |
|---|---|---|---|---|
| Transketolase | Pentose Phosphate Pathway | TktA, TktB | 15% | Reduced flux significantly impacts pentose phosphate pathway function |
| TCA cycle reactions | Tricarboxylic Acid Cycle | Multiple | 19% | Limited robustness to flux variations |
| Ribose-5-phosphate isomerase | Pentose Phosphate Pathway | RpiA, RpiB | 28% | Moderate robustness to flux reduction |
| Three-carbon glycolytic reactions | Glycolysis | GapA, Pgk, etc. | Limited robustness | Both increased and decreased fluxes affect growth predictions significantly |
Data adapted from Edwards et al. [56]
PTS Mutant Phenotypes and Growth Characteristics
Strategic manipulation of PTS components has yielded mutant strains with altered growth characteristics that highlight the discrepancies between predicted and actual metabolic performance:
Table 2: PTS Mutant Strains and Their Metabolic Characteristics
| Strain Type | Engineering Approach | Growth Characteristics | Carbon Utilization | Acetate Production |
|---|---|---|---|---|
| PTS(-) glc(+) | Adaptive selection in chemostat | Restored growth on glucose | Simultaneous sugar consumption | Reduced |
| PTS(-) with recombinant transporters | GalP promoter replacement or recombinant glucose transport | Improved growth on glucose | Enhanced glucose uptake | Variable |
| crp+ enhanced mutants | Enhanced Crp expression in mgsA, pgi, and ptsG mutants | Abolished CCR | Simultaneous consumption of mixed sugars | Low |
Data summarized from Escalante et al. [52]
Experimental Protocols
Protocol 1: Model Refinement for PTS Representation
Purpose: To enhance the accuracy of FBA growth predictions by improving PTS and central metabolism annotations in metabolic models.
Materials:
- EColiCore2 or genome-scale model (iJO1366)
- COBRA Toolbox or similar constraint-based modeling environment
- Biochemical literature on PTS components and interactions
Procedure:
- Model Selection and Import:
- Obtain EColiCore2 model from systemsbiology.ucsd.edu [57] or iJO1366 from BiGG Models database
- Import model into COBRA Toolbox using
readCbModelfunction
PTS Component Verification:
- Confirm presence of all major PTS sugar branches (glucose, mannose, N-acetylglucosamine)
- Validate phosphotransfer reactions from PEP to EI, HPr, and EII components
- Check for nitrogen-related PTS components (EINtr, NPr, EIIANtr) based on recent research [54]
Regulatory Constraint Implementation:
- Incorporate catabolite repression rules using Boolean regulatory functions
- Apply flux constraints based on PTS phosphorylation state
- Implement inducer exclusion mechanisms for non-PTS sugars
Network Reduction (Optional):
- Use NetworkReducer algorithm [55] to derive core models preserving PTS functions
- Specify protected phenotypes: growth on glucose, mannose, and other PTS sugars
- Maintain key anaplerotic reactions and electron transport chain components
Validation and Testing:
- Simulate growth on single PTS sugars and compare with experimental data
- Test diauxic growth predictions on sugar mixtures
- Perform robustness analysis on essential PTS reactions
Protocol 2: Experimental Validation of PTS Function
Purpose: To experimentally characterize PTS activity and validate computational predictions of growth parameters.
Materials:
- E. coli wild-type and PTS mutant strains
- FRET-compatible fluorescent protein tags (CFP, YFP)
- M9 minimal media with various carbon sources
- Microplate reader or fluorometer for growth and FRET measurements
Procedure:
- Strain Preparation:
- Clone PTS components (EI, HPr, EIIA, EIIBC) with C-terminal CFP/YFP tags
- Transform plasmids into appropriate E. coli strains
- Verify expression via Western blot or fluorescence measurement
FRET-based Interaction Monitoring:
- Grow cells to mid-log phase in minimal media with non-PTS carbon source
- Transfer aliquots to microplate for fluorescence readings
- Measure baseline CFP and YFP emissions (excite at 433 nm, detect at 475 nm and 527 nm)
- Add PTS sugars at varying concentrations (0.1-10 mM)
- Monitor FRET ratio (YFP/CFP emission) over time [53]
Growth Phenotype Characterization:
- Inoculate wild-type and mutant strains into media with single and mixed sugars
- Measure OD600 every 30 minutes for 24-48 hours
- Calculate growth rates and lag phases for each condition
- Identify diauxic shifts in mixed sugar conditions
Data Integration with Metabolic Models:
- Incorporate measured uptake rates as constraints in FBA
- Compare predicted vs. actual growth rates and substrate preferences
- Refine model parameters based on discrepancies
The Scientist's Toolkit
Table 3: Essential Research Reagents for PTS and Metabolic Studies
| Reagent/Resource | Type | Function/Application | Example Sources |
|---|---|---|---|
| EColiCore2 Model | Computational Resource | Core metabolic model for FBA of central metabolism | UCSD Systems Biology [57] |
| COBRA Toolbox | Software Package | MATLAB toolbox for constraint-based modeling | opencobra.github.io |
| iJO1366 Model | Computational Resource | Genome-scale E. coli metabolic reconstruction | BiGG Models database |
| NetworkReducer | Algorithm | Network reduction while preserving phenotypes | Büchel et al. [55] |
| FRET-compatible Fluorescent Proteins | Experimental Reagent | Monitoring protein interactions in live cells | CFP/YFP tagged PTS components [53] |
| PTS Mutant Strains | Biological Material | Studying PTS functions and regulatory roles | Various mutant collections [52] |
PTS Signaling Pathway Diagram
Addressing growth prediction errors in E. coli metabolic models requires careful attention to PTS annotation and central metabolism representation. The integration of computational refinements through model reduction techniques like EColiCore2 and experimental validation using FRET-based interaction studies provides a robust framework for improving FBA accuracy. The protocols and analyses presented here offer researchers a structured approach to bridge the gap between theoretical metabolic predictions and experimental observations, ultimately enhancing the reliability of in silico models for metabolic engineering and drug development applications.
Calibrating Model Constraints Using Experimental Data on PTS Protein Phosphorylation States
The phosphotransferase system (PTS) is a major carbohydrate transport system in bacteria such as Escherichia coli, performing vectorial phosphorylation of sugars using phosphoenolpyruvate (PEP) as the phosphoryl donor [24] [58]. Beyond transport, the phosphorylation states of PTS proteins form a central phosphorylation cascade that regulates carbon metabolism, including phenomena like carbon catabolite repression (CCR) and inducer exclusion [24] [59]. For constraint-based metabolic models, such as those used in Flux Balance Analysis (FBA), accurately representing the regulatory constraints imposed by the PTS is essential for predicting bacterial physiological responses, particularly when dealing with mutant strains or different carbon sources [60] [61]. This application note details how quantitative experimental data on PTS protein phosphorylation states and mutant phenotypes can be leveraged to calibrate and validate such models, creating a more robust framework for predicting metabolic behavior.
Quantitative Experimental Data for Model Constraints
Physiological Characterization of PTS Mutant Strains
Data from defined PTS knockout strains provides a direct means to parameterize and validate model constraints. The following table summarizes key physiological parameters for E. coli W3110 derivative strains with impairments in their glucose uptake systems, cultivated under batch conditions with excess glucose [60].
Table 1: Physiological Parameters of E. coli PTS Glucose Uptake Mutant Strains
| Strain & Genotype | Description | Max. Specific Growth Rate (μmax, h⁻¹) | Max. Specific Glucose Uptake Rate (qSmax, mmol/gCDW/h) | Max. Specific Acetate Production Rate (qApmax, mmol/gCDW/h) | Biomass Yield on Glucose (YXS, gCDW/mol) |
|---|---|---|---|---|---|
| Wild-Type (W3110) | Reference strain | 0.41 | 8.5 | 5.8 | 55.2 |
| WG (ΔptsG) | Lacks the glucose-specific EIIBC component | 0.32 (78% of WT) | 5.1 (60% of WT) | 2.0 (34% of WT) | 58.1 |
| WGP (ΔptsG, ΔgalP) | Lacks EIIBCGlc and the galactose:H+ symporter GalP | 0.31 (76% of WT) | 4.9 (58% of WT) | 1.9 (33% of WT) | 57.9 |
| WGM (ΔptsG, ΔmanX) | Lacks EIIBCGlc and the mannose-specific EIIAB component | 0.23 (56% of WT) | 3.2 (38% of WT) | Not Detected | 63.5 |
Phosphorylation State Data and Regulatory Output
The phosphorylation state of EIIAGlc is a key regulatory signal in the PTS. Its dephosphorylated form accumulates during active PTS sugar transport and inhibits non-PTS carbohydrate uptake systems (inducer exclusion), while its phosphorylated form activates adenylate cyclase, influencing global gene expression via cAMP-CRP [24] [59]. The degree of phosphorylation of EIIAGlc can be mathematically related to external stimuli.
Table 2: Key Quantitative Relationships for PTS Signaling
| Parameter / Relationship | Description | Mathematical Expression / Value | Experimental Context |
|---|---|---|---|
| Phosphorylation State Steady-State | EIIAGlc phosphorylation depends on the intracellular PEP:pyruvate ratio and extracellular PTS sugar concentration. | A characteristic field describes the fraction of phosphorylated EIIAGlc as a function of [Sucrose]ex and [PEP]:[Pyruvate] [59]. | Determined in chemostat cultures; the phosphorylation state can be measured via Western blot or enzyme assays. |
| Dynamic Response Time | The phosphate transfer through the PTS cascade occurs rapidly. | The system reaches a new steady-state within ~1 second after a perturbation [59]. | Measured in stimulus-response experiments. |
| Critical Metabolite Ratio | The intracellular ratio of PEP to pyruvate is a central energy marker that drives phosphorylation. | Varies with carbon source and metabolic state; a high ratio favors the phosphorylated state of PTS proteins [59]. | Can be quantified using enzymatic assays or LC-MS/MS on ethanol-boiling extracts. |
Experimental Protocols for Key Determinations
Protocol 1: Cultivation of PTS Mutant Strains and Determination of Physiological Parameters
This protocol outlines the cultivation of E. coli PTS mutant strains and the measurement of growth parameters for metabolic model validation [60].
Strains and Pre-Cultivation:
- Use defined E. coli K-12 knockout strains (e.g., WG, WGM, WGP).
- Inoculate 15 mL of TY medium (16 g/L tryptone, 10 g/L yeast extract, 5 g/L NaCl) from a cryo-stock. Incubate for 7 h at 37°C and 220 rpm.
- Use the first pre-culture to inoculate a second pre-culture in a mineral salt medium (MSM) supplemented with 25% (v/v) TY medium to an OD600 of 0.25. Incubate for 12 h.
Main Cultivation and Monitoring:
- Inoculate the main bioreactor (MSM with defined glucose concentration, e.g., 10-20 g/L) to an initial OD600 of 0.5.
- Cultivate in a batch system with controlled temperature (37°C), pH (7.0), and dissolved oxygen.
- Monitor growth by measuring OD600 offline or online.
- Take periodic samples for substrate (glucose) and metabolite (acetate) analysis via HPLC.
Parameter Calculation:
- Calculate the maximum specific growth rate (μmax) by fitting the exponential phase of the growth curve.
- Determine the maximum specific substrate uptake rate (qSmax) and maximum specific acetate production rate (qApmax) using the measured concentrations, biomass data, and the known growth rate.
- Calculate the biomass yield (YXS) as the total biomass produced per mol of glucose consumed.
Protocol 2: Quantifying the Phosphorylation State of EIIAGlc
This protocol describes a method to determine the phosphorylation state of EIIAGlc, a critical regulatory variable [59].
Culture in a Chemostat:
- Grow an E. coli strain in a continuously stirred tank reactor (CSTR) under carbon-limiting conditions (e.g., sucrose limitation) at a fixed dilution rate.
- Vary the input signal by changing the dilution rate (affecting the intracellular PEP:pyruvate ratio) or the feed substrate concentration.
Rapid Sampling and Metabolite Quenching:
- Rapidly withdraw culture samples.
- Immediately quench metabolism using a boiling ethanol-buffer solution (60% ethanol, 40% 20 mM HEPES, pH 7.5) at 80°C to freeze the metabolic state instantaneously.
Sample Analysis:
- Intracellular Metabolites: Analyze the concentrations of PEP and pyruvate in the quenched and extracted samples using enzymatic assays or LC-MS/MS.
- EIIAGlc Phosphorylation State: Determine the fraction of phosphorylated EIIAGlc using non-denaturing gel electrophoresis followed by Western blotting with specific antibodies, or via functional enzyme assays that distinguish between the phosphorylated and unphosphorylated forms.
Integration into a Constraints-Based Metabolic Model
Model Structure and Integration Workflow
The process of integrating experimental PTS data into a metabolic model can be visualized as a cyclic workflow of simulation, experimental validation, and model calibration. The core structure of the PTS is a phosphorylation cascade that can be represented in a stoichiometric model.
Diagram 1: PTS cascade with regulatory outputs and model calibration workflow.
Implementing Constraints and Validating the Model
- Reaction Constraints: Incorporate the PTS transport reaction for specific sugars (e.g., glucose) as a group translocation reaction consuming PEP and producing the corresponding sugar-phosphate and pyruvate [59]. The stoichiometry must be accurately represented.
- Regulatory Constraints: Implement the regulatory effects of EIIAGlc on other transporters. For example, the flux through a non-PTS transporter (e.g., lactose) can be constrained to zero when EIIAGlc is predominantly dephosphorylated, a state that occurs during rapid glucose transport [61] [59]. This can be achieved with Boolean logic or thermodynamic constraints in a metabolic-regulatory model [61].
- Parameterization and Validation:
- Use the physiological data from Table 1 to set upper bounds for glucose uptake in different mutant strains (e.g., lower qSmax for WG and WGM strains).
- Validate the calibrated model by testing its ability to predict the growth rates and acetate production profiles of the mutant strains under conditions not used for parameterization.
- Compare the simulated flux distributions and carbon usage priorities with experimental observations, such as the absence of acetate overflow in the WGM strain.
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Reagents and Resources for PTS and Constraint-Based Modeling Research
| Reagent / Resource | Function / Application | Specifications / Examples |
|---|---|---|
| Defined E. coli KO Strains | Genetically engineered hosts for validating model predictions of PTS function and regulation. | E. coli WG (ΔptsG), WGM (ΔptsG, ΔmanX), WGP (ΔptsG, ΔgalP) [60]. |
| Mineral Salt Medium (MSM) | Defined cultivation medium for controlled experiments and accurate quantification of metabolic fluxes. | Contains (NH4)2SO4, K2HPO4/NaH2PO4 buffer, trace elements, MgSO4, thiamine, and a defined carbon source [60]. |
| Parallel Bioreactor Systems | High-throughput cultivation with enhanced process control for generating reproducible physiological data. | 10 mL mini-bioreactors integrated with Liquid Handling Stations; 100 mL small-scale bioreactors with off-gas analysis for respiration data [60]. |
| Quenching Solution (Boiling Ethanol) | Rapid quenching of intracellular metabolism to capture the in vivo state of metabolites and protein phosphorylation. | 60% Ethanol, 40% 20 mM HEPES buffer, pH 7.5, maintained at 80°C [59]. |
| Genome-Scale Metabolic Model | In silico platform for simulating metabolism and integrating experimental data to predict physiological states. | E. coli K-12 model iML1515 [62] or other relevant reconstructions. Can be expanded to include transcriptional regulation [61]. |
| PTSD | Database and resource for information on PTS proteins, their sequences, and known functions across bacteria. | Contains information on PTS protein-encoding genes and their classification [58]. |
The phosphoenolpyruvate (PEP)-carbohydrate phosphotransferase system (PTS) represents a crucial gateway for carbon uptake in Escherichia coli and many other bacteria [52] [63]. This system couples the transport and phosphorylation of sugars such as glucose using PEP as the phosphoryl donor and energy source [15]. The general PTS proteins, Enzyme I (encoded by ptsI) and the histidine phosphocarrier protein (HPr, encoded by ptsH), are essential for the phosphorylation cascade that ultimately transfers the phosphoryl group to incoming sugars via substrate-specific enzyme II complexes [64] [65]. Knocking out ptsH or ptsI genes produces profound phenotypic consequences that extend beyond the inability to transport PTS sugars, affecting central carbon metabolism, regulatory networks, and cellular physiology [64] [66] [52]. This protocol details strategies for modeling these knockouts and interpreting their complex phenotypes within constraint-based modeling frameworks, particularly Flux Balance Analysis (FBA).
Background: Physiological Impact of ptsH/ptsI Knockouts
Core PTS Function and Knockout Consequences
The PTS phosphorylation chain begins with PEP-dependent autophosphorylation of Enzyme I, which subsequently transfers the phosphoryl group to HPr. Phospho-HPr then phosphorylates various sugar-specific EIIA components, which ultimately transfer the phosphoryl group to the incoming sugar during its translocation through the corresponding EIIC membrane transporter [15] [63]. Knockouts of ptsI or ptsH disrupt this entire phosphorylation cascade, rendering cells unable to transport and phosphorylate PTS sugars [64] [65].
Extended Phenotypic Effects
Beyond the expected inability to grow on PTS sugars, ptsH and tight ptsI mutants exhibit pleiotropic effects including:
- Inability to utilize certain non-PTS sugars (maltose, melibiose, glycerol, lactose) due to PTS-mediated repression [64] [65] [52]
- Altered carbon catabolite repression (CCR) and deregulated induction of catabolic enzyme systems [64] [52]
- Reduced proton motive force (PMF) and reactive oxygen species (ROS) generation, contributing to antibiotic resistance against β-lactams and aminoglycosides [67]
- Metabolic network rewiring evidenced by flux rerouting through alternative pathways and changes in metabolite concentrations [68] [66]
Table 1: Key Phenotypic Differences Between ptsH and ptsI Mutants
| Phenotypic Characteristic | ptsI Mutants | ptsH Mutants | Experimental Reference |
|---|---|---|---|
| Growth on fructose | Unable | Able | [64] [65] |
| Utilization of other PTS sugars after growth on fructose | Unable | Able for few generations | [64] [65] |
| Relief of PTS-mediated repression after growth on fructose | No relief | Significant relief | [64] [65] |
| Requirement for secondary mutations for glucose utilization | Essential | Less critical | [66] [52] |
Experimental Characterization Protocols
Physiological Characterization of PTS Knockouts
Growth Phenotyping
Objective: Quantify growth capabilities of knockout strains under various carbon conditions.
Materials:
- Bacterial Strains: Keio collection knockout strains (e.g., JW2410-1 for crr, JW1087-2 for ptsG) or constructed ptsH/ptsI knockouts in desired background (e.g., BW25113) [68]
- Growth Media: M9 minimal medium supplemented with 2 g/L of various carbon sources (glucose, fructose, maltose, lactose, glycerol, etc.) [68]
- Equipment: Microbioreactor system or shaking incubator with flask, spectrophotometer for OD600 measurements [68] [69]
Procedure:
- Inoculate single colonies into 5 mL LB medium and grow overnight at 37°C with shaking.
- Wash cells twice with sterile M9 salts to remove residual carbon.
- Dilute cultures to OD600 ≈ 0.05 in M9 minimal medium containing specific carbon sources.
- Monitor growth by measuring OD600 every 30-60 minutes until stationary phase.
- Calculate maximum growth rate (μmax) from the exponential phase and final biomass yield.
Expected Results: ptsH/ptsI knockouts should show no growth on glucose and other PTS sugars, potentially impaired growth on some non-PTS sugars, and possible growth on non-PTS carbon sources such as glycerol or maltose depending on the strain background and specific regulatory adaptations [64] [52].
Metabolic Flux Analysis
Objective: Quantify intracellular metabolic fluxes in knockout strains.
Materials:
- Isotope Labeling: [U-13C]glucose (Cambridge Isotope Laboratories) [68]
- Analytical Equipment: GC-MS or LC-MS systems for metabolite detection, HPLC system for extracellular metabolite analysis [68]
- Culture System: Aerated mini-bioreactors with 10 mL working volume or controlled fermenters [68]
Procedure:
- Grow knockout and control strains in M9 minimal medium containing [U-13C]glucose.
- Harvest cells during mid-exponential phase (OD600 ≈ 0.7).
- Quench metabolism rapidly using cold methanol.
- Extract intracellular metabolites and analyze mass isotopomer distributions.
- Measure extracellular metabolite concentrations (organic acids, residual sugars).
- Calculate metabolic fluxes using computational tools such as INCA or OpenFlux, constrained by measured uptake and secretion rates.
Expected Results: PTS knockouts typically show redirected carbon flux through alternative pathways such as the Entner-Doudoroff pathway and pentose phosphate pathway, reduced glycolytic flux, and potentially altered TCA cycle activity [68] [66].
Molecular Characterization
Transcriptomic Analysis
Objective: Identify global gene expression changes in response to PTS knockout.
Procedure:
- Extract total RNA from mid-exponential phase cultures using standard kits.
- Prepare RNA sequencing libraries and perform high-throughput sequencing.
- Map reads to reference genome and quantify gene expression levels.
- Perform differential expression analysis comparing knockout to wild-type.
- Conduct pathway enrichment analysis to identify affected biological processes.
Expected Results: Upregulation of alternative carbon catabolic pathways, changes in stress response genes, and alterations in carbon catabolite repression targets [66] [69].
Computational Modeling Strategies
Constraint-Based Modeling of PTS Knockouts
Flux Balance Analysis (FBA)
Objective: Predict metabolic behavior of PTS knockout strains.
Procedure:
- Model Preparation: Use a genome-scale metabolic model of E. coli (e.g., iJO1366).
- Knockout Implementation: Set fluxes through reactions catalyzed by PTS enzymes to zero.
- Constraint Definition: Apply measured substrate uptake rates as constraints.
- Objective Function: Typically maximize biomass production.
- Simulation: Solve the linear programming problem to predict flux distributions.
Limitations: Standard FBA often fails to accurately predict PTS knockout phenotypes due to regulatory constraints not captured in stoichiometric models [68].
Advanced Constraint-Based Approaches
Minimization of Metabolic Adjustment (MOMA)
- Principle: Assumes the knockout strain minimizes the Euclidean distance between wild-type and mutant flux distributions [68]
- Application: More accurate for predicting immediate metabolic responses to gene knockouts
- Implementation: Solve quadratic programming problem minimizing ||v mutant - v wild-type||²
Regulatory On/Off Minimization (ROOM)
- Principle: Minimizes the number of significant flux changes relative to wild-type
- Application: Suitable for modeling adaptive evolution where regulatory networks have been optimized
Relative Optimality in Metabolic Networks (RELATCH)
- Principle: Incorporates gene expression data from reference state to constrain flux predictions
- Application: Useful when transcriptomic data are available for the knockout strain
Table 2: Comparison of Modeling Approaches for PTS Knockouts
| Method | Theoretical Basis | Advantages | Limitations | Accuracy for PTS Knockouts |
|---|---|---|---|---|
| FBA | Maximizes biomass production | Simple, fast | Poor prediction of knockout phenotypes | Low [68] |
| MOMA | Minimizes Euclidean distance from wild-type flux | Better for immediate knockout response | Does not incorporate regulation | Moderate [68] |
| ROOM | Minimizes number of flux changes | Captures regulatory constraints | Requires wild-type reference | Moderate to High |
| RELATCH | Incorporates gene expression data | Uses multi-omics data | Requires additional experimental data | High [68] |
Integrating Multi-omics Data
Objective: Improve model predictions by incorporating experimental data.
Procedure:
- Transcriptomic Integration: Use methods like E-Flux or PROM to constrain flux bounds based on gene expression data.
- Metabolomic Integration: Incorporate measured metabolite pool sizes as additional constraints.
- Flomic Integration: Use 13C-MFA derived flux measurements to validate and refine model predictions.
- Iterative Refinement: Compare predictions with experimental data and adjust model constraints accordingly.
Expected Outcome: More accurate prediction of metabolic states in PTS knockouts, including flux rerouting and identification of compensatory mechanisms [66].
The Scientist's Toolkit: Research Reagent Solutions
Table 3: Essential Research Reagents for PTS Knockout Studies
| Reagent/Category | Specific Examples | Function/Application | Key References |
|---|---|---|---|
| Bacterial Strains | Keio collection knockouts (JW2410-1, JW1087-2); BW25113 background strains | Provide defined genetic background for phenotypic studies | [68] |
| Carbon Sources | [U-13C]glucose; various PTS and non-PTS sugars | Metabolic flux analysis; growth phenotyping | [68] |
| Culture Systems | Aerated mini-bioreactors; microbioreactor arrays | Controlled growth conditions for physiological characterization | [68] [69] |
| Analytical Tools | GC-MS; HPLC systems | Metabolite detection and quantification | [68] |
| Computational Models | iJO1366 genome-scale model; constraint-based modeling tools | Prediction of metabolic behavior | [68] [66] |
| Molecular Biology Tools | RNA sequencing kits; plasmid vectors for complementation | Molecular characterization and validation | [66] [69] |
Signaling and Metabolic Pathways
The PTS network operates as both a transport system and a regulatory hub, with ptsH and ptsI knockouts causing widespread disruption to carbon metabolism and gene regulation. The following diagram illustrates the central position of these genes in the metabolic network and the cascading effects of their knockout:
Diagram Title: PTS Knockout Network Effects
Experimental-Computational Workflow
The following diagram outlines an integrated approach combining experimental characterization and computational modeling for comprehensive analysis of PTS knockouts:
Diagram Title: PTS Analysis Workflow
Troubleshooting and Validation
Common Challenges in PTS Knockout Modeling
Regulatory Effects Overshadow Stoichiometric Constraints
- Problem: Traditional FBA fails due to strong regulatory constraints not captured in models
- Solution: Use MOMA or ROOM for immediate knockout response prediction [68]
Adaptive Mutations Complicate Prediction
- Problem: Evolved strains accumulate compensatory mutations that alter physiology [66]
- Solution: Compare unevolved and evolved knockouts to distinguish direct and indirect effects
Carbon Source-Dependent Effects
Model Validation Approaches
- Flux Validation: Compare predicted fluxes with 13C-MFA measurements [68] [66]
- Growth Prediction Validation: Compare predicted growth rates/yields with experimental measurements
- Gene Essentiality Validation: Test model predictions of essential genes against experimental data
Modeling PTS knockouts requires integrated experimental and computational approaches that account for both stoichiometric constraints and regulatory influences. The pleiotropic effects of ptsH and ptsI knockouts stem from their central role in carbon metabolism and regulation, necessitating multi-omic characterization and advanced modeling techniques beyond standard FBA. The protocols outlined here provide a framework for comprehensive analysis of PTS knockout strains, enabling more accurate prediction of their metabolic behavior and supporting applications in metabolic engineering and systems biology research.
The phosphoenolpyruvate (PEP)-carbohydrate phosphotransferase system (PTS) constitutes a primary carbohydrate uptake mechanism in Escherichia coli and numerous other bacteria [52]. This system concurrently transports and phosphorylates its sugar substrates, utilizing PEP as the phosphoryl donor. A significant metabolic constraint arises because glucose transport through the PTS consumes one molecule of PEP per molecule of glucose internalized, thereby depleting the intracellular pool of PEP [52]. This pool is vital as a precursor for the biosynthesis of commercially significant aromatic compounds, antimicrobials, and platform chemicals. Consequently, the inherent function of the PTS creates a direct competition between sugar uptake and the synthesis of valuable metabolites.
Inactivation of the PTS presents a strategic approach to decouple sugar uptake from PEP consumption, thereby increasing the availability of PEP for biosynthetic purposes [52]. This application note details the underlying rationale, experimental methodologies, and computational protocols for implementing PTS inactivation in E. coli to enhance PEP supply. The content is framed within a broader thesis on employing Flux Balance Analysis (FBA) to analyze and optimize such metabolic interventions.
Background and Rationale
The PTS and its Role in Central Carbon Metabolism
The PTS is a multi-component system that facilitates the vectorial phosphorylation of sugars during their transport across the cytoplasmic membrane. In E. coli, the glucose-specific PTS involves a cascade of phosphotransfer reactions from PEP to the transmembrane transporter IICBGlc via the general proteins Enzyme I (EI) and HPr, and the sugar-specific component IIAGlc [15]. The terminal step involves the phosphorylation of the incoming glucose molecule by the transporter, yielding intracellular glucose-6-phosphate and pyruvate [52] [15]. This mechanism positions the PTS at the intersection of carbohydrate uptake and central carbon metabolism, with an estimated 50% of the PEP pool being consumed by the PTS during growth on glucose [52].
A critical discovery with profound implications for metabolic engineering is the demonstrated reversibility of the Enzyme I-catalyzed reaction. Recent in vivo studies using 13C tracer analysis have confirmed that EI facilitates a significant flux from pyruvate back to PEP, not only during gluconeogenic growth but also during growth on glycolytic substrates like glucose [70]. This reverse flux, quantified at approximately 10% of the PEP pool during growth on glucose, challenges the traditional view of the PEP-to-pyruvate step as irreversible and reveals a previously unrecognized capacity of the PTS to contribute to PEP regeneration [70].
PTS Inactivation for Metabolic Engineering
The deletion of PTS genes (creating PTS- mutants) seeks to eliminate the consumption of PEP for sugar phosphorylation. This strategy aims to increase the intracellular concentration of PEP, thereby augmenting the flux into pathways that use PEP as a precursor, such as the aromatic amino acid pathway [52]. However, a major challenge is that PTS- mutants typically lose the ability to transport and grow on PTS sugars like glucose. A key objective of the associated protocols is, therefore, to generate PTS- mutants that have regained the ability to utilize glucose (PTS- glc+), often through the activation of alternative, non-PTS uptake systems [52].
Table 1: Key Metabolic Engineering Strategies for PTS Inactivation and Their Outcomes
| Strategy | Genetic Modifications / Selection Method | Key Physiological Outcomes | Relevant Citations |
|---|---|---|---|
| Adaptive Evolution | Adaptive selection in chemostats on glucose. | Selection of PTS- glc+ mutants; simultaneous consumption of mixed carbon sources; reduced acetate excretion. | [52] |
| Promoter Engineering & Transporter Expression | Replacement of native galP promoter with strong constitutive promoters; expression of recombinant glucose transporters (e.g., glk). | Restored glucose uptake via GalP proton symport and phosphorylation by glucokinase; increased PEP pool. | [52] [70] |
| CRP Enhancement | Overexpression of the cAMP receptor protein (Crp). | Abolishment of carbon catabolite repression (CCR); enables co-utilization of sugar mixtures; reduces acetate production. | [52] |
Computational Analysis via Flux Balance Analysis (FBA)
FBA Protocol for Simulating PTS Inactivation
Flux Balance Analysis is a constraint-based modeling approach used to predict the flow of metabolites through a metabolic network at steady state [71] [72]. The following protocol outlines its application for analyzing the metabolic consequences of PTS inactivation.
Protocol 1: In Silico Simulation of PTS Gene Deletion using FBA
- Model Selection and Import: Obtain a genome-scale metabolic model of E. coli (e.g., iJO1366). Load the model into a constraint-based modeling environment such as the COBRA Toolbox for MATLAB/Python or a similar platform.
- Define Baseline Constraints: Set the upper and lower bounds of exchange reactions to define the growth medium (e.g., minimal glucose medium). The glucose uptake rate is typically constrained to a value like 10 mmol/gDW/h.
- Set the Objective Function: Define the biomass reaction as the objective function to be maximized, simulating rapid growth.
- Simulate Wild-Type Flux Distribution: Perform an FBA simulation to compute the wild-type growth rate and flux distribution. This serves as a control.
- Implement PTS Gene/Reaction Deletion:
- Identify the reactions associated with the target PTS genes (ptsG, ptsI, crr). For ptsG (encoding IICBGlc), this is the glucose PTS transport reaction.
- To simulate a knockout, constrain the flux through the corresponding reaction(s) to zero.
- For a ptsI (EI) knockout, note that this disrupts all PTS sugar uptake. The model may predict no growth on glucose unless an alternative uptake route is specified.
- Simulate Mutant Phenotype: Re-run the FBA with the new constraints. The model will predict a new growth rate and flux distribution.
- Analyze Results:
- Growth Rate: Compare the mutant's predicted growth rate (μ) to the wild type.
- PEP Pool Analysis: Examine the flux values for reactions consuming and producing PEP.
- Byproduct Secretion: Check for changes in acetate, formate, or other byproduct secretion fluxes.
- Flac Variability Analysis (FVA): Perform FVA on the mutant model to determine the range of possible fluxes for the PEP carboxylase (PPC) or other key precursor-generating reactions, assessing the network's capacity to supply precursors.
This in silico analysis can identify seven gene products of central metabolism essential for aerobic growth on glucose and 15 for anaerobic growth, providing a predictive framework before experimental work [71].
Visualizing the FBA Workflow for PTS Analysis
The following diagram illustrates the logical workflow for applying FBA to analyze PTS inactivation, from model setup to result interpretation.
Experimental Protocols
Generation of PTS-glc+Strains
Protocol 2: Adaptive Laboratory Evolution (ALE) for Selecting PTS- glc+ Mutants
Principle: Serial passaging or continuous culture in a glucose-limited environment selects for spontaneous mutants that have acquired mutations allowing them to utilize glucose via non-PTS mechanisms [52].
Materials:
- Strain: E. coli K-12 MG1655 ΔptsG (or other PTS-null mutant).
- Equipment: Chemostat or biological reactors for batch culture.
- Media: M9 minimal medium with glucose (e.g., 2 g/L) as the sole carbon source.
Method:
- Inoculation: Inoculate the PTS- strain into a chemostat or serial batch culture containing the glucose minimal medium.
- Culture Conditions: Maintain the culture under constant dilution rate (in a chemostat) or through repeated transfers during the late exponential phase (in batch). The low glucose concentration imposes a strong selective pressure.
- Monitoring: Monitor culture density (OD600) over time. An initial lag period is expected, followed by a gradual increase in growth rate and culture density as beneficial mutations arise and enrich.
- Isolation: After a significant increase in fitness is observed (typically over weeks), plate culture samples onto minimal glucose agar to isolate single colonies.
- Validation: Screen individual clones for their ability to grow robustly on glucose minimal medium. Confirm the PTS- genotype (e.g., via PCR) and the PTS- glc+ phenotype.
Protocol 3: Rational Engineering of a Glucose Uptake System in a PTS- Strain
Principle: The native galactose permease (GalP) can transport glucose, and glucokinase (Glk) can phosphorylate it. Enhancing the expression of these proteins provides a direct route to restore glucose utilization without the PTS [52] [70].
Materials:
- Strain: E. coli PTS- strain (e.g., ΔptsHIcrr or ΔptsG).
- Plasmids: Constructs containing galP and glk under the control of strong, constitutive promoters (e.g., Plac, Ptrc).
Method:
- Genetic Modification: Replace the native promoter of the galP gene with a strong promoter using λ-Red recombineering or CRISPR-Cas9. Alternatively, introduce a plasmid expressing both galP and glk.
- Transformation: Transform the engineered construct into the PTS- strain.
- Selection and Cultivation: Select transformants on minimal agar plates containing glucose as the sole carbon source.
- Characterization: Quantify the growth rate, glucose uptake rate, and acetate production in shake-flask cultures. Compare these metrics to the wild-type and unevolved PTS- strains.
Quantifying Metabolic Fluxes
Protocol 4: Measuring PYR→PEP Reverse Flux via 13C Tracer Analysis
Principle: This protocol uses [U-13C]alanine, which equilibrates with the intracellular pyruvate pool, to quantify the flux from pyruvate (PYR) back to PEP, a flux facilitated by EI and PpsA [70].
Materials:
- Strains: Wild-type E. coli, ΔppsA, ΔptsI, and ΔppsAΔptsI.
- Tracer: [U-13C]alanine.
- Equipment: GC-MS (Gas Chromatography-Mass Spectrometry) or LC-MS (Liquid Chromatography-Mass Spectrometry).
- Media: Defined minimal medium with a desired carbon source (e.g., glucose, acetate).
Method:
- Cultivation: Grow cultures in minimal medium to mid-exponential phase.
- Tracer Pulses: Add [U-13C]alanine directly to the culture. Allow metabolism to proceed for a short, defined period (e.g., 30 seconds to a few minutes).
- Quenching and Metabolite Extraction: Rapidly quench metabolism (e.g., using cold methanol) and extract intracellular metabolites.
- Sample Analysis:
- Derivatize proteinogenic amino acids from the biomass hydrolysate.
- Analyze the samples using GC-MS. PYR labeling is observed via valine, OAA via aspartate, and PEP via phenylalanine.
- Data Interpretation:
- Calculate the mass isotopomer distributions (MIDs) for the measured fragments.
- Use the labeling patterns to determine the relative contribution of PYR to PEP. The presence of M1 labeling in PEP (from [U-13C]alanine) indicates a direct flux from the labeled PYR pool.
Table 2: Quantitative Analysis of PYR→PEP Flux in Different Genetic Backgrounds [70]
| Strain Genotype | Growth on Acetate | Growth on Glucose | PYR→PEP Flux on Acetate | PYR→PEP Flux on Glucose |
|---|---|---|---|---|
| Wild Type | Normal | Normal | ~60% | ~10% |
| ΔppsA | Normal | Normal | ~65% | ~10% |
| ΔptsI | Normal | Very Slow (requires adaptation) | ~65% | N/D |
| ΔppsA ΔptsI | Unable to grow on PYR; impaired on acetate | Very Slow (requires adaptation) | ~0% | ~2.4% |
The Scientist's Toolkit
Table 3: Essential Research Reagent Solutions for PTS and Metabolic Flux Studies
| Reagent / Tool | Function / Description | Application in PTS Research |
|---|---|---|
| Genome-Scale Model (GEM) | A computational reconstruction of an organism's metabolism (e.g., E. coli iJO1366). | Predicting metabolic fluxes, growth phenotypes, and outcomes of gene knockouts in silico [71] [72]. |
| Flux Balance Analysis (FBA) | A mathematical method to simulate metabolism by optimizing an objective function (e.g., growth) under stoichiometric constraints. | Analyzing the theoretical capabilities of PTS-modified strains and identifying optimal genetic interventions [71] [72]. |
| 13C Isotopic Tracers | Stable isotope-labeled compounds (e.g., [1-13C]glucose, [U-13C]alanine). | Experimentally quantifying intracellular metabolic fluxes, including non-obvious routes like EI-mediated reverse flux [70]. |
| CRISPR-Cas9 / λ-Red Recombineering | Genome editing tools for precise genetic modifications. | Constructing PTS knockout mutants (e.g., ΔptsG, ΔptsI) and engineering alternative transporters [52]. |
| GC-MS / LC-MS | Analytical instruments for separating and identifying chemical compounds. | Measuring the incorporation of 13C labels into metabolites to determine flux distributions [70]. |
Integrated Pathway and Workflow Visualization
The following diagram synthesizes the core concepts, strategies, and outcomes of leveraging PTS inactivation to boost PEP supply, integrating the roles of key enzymes and engineering strategies.
Carbon Catabolite Repression (CCR) is a fundamental global control system in bacteria that allows them to prioritize preferred carbon sources when multiple substrates are available [73]. In Escherichia coli, this system ensures that glucose is utilized first before secondary carbon sources such as lactose, creating characteristic diauxic growth patterns [74] [73]. The molecular mechanisms governing this process involve complex interactions across metabolism, gene expression, and signaling pathways, subject to global physical and physiological constraints [74].
A key mechanism within CCR is inducer exclusion, where the transport and metabolism of preferred carbon sources directly inhibit the uptake of non-preferred substrates [75] [76]. In E. coli, this process is primarily mediated through the phosphotransferase system (PTS), a multicomponent system responsible for the transport and phosphorylation of various sugars [75] [73]. Understanding and accurately modeling these regulatory mechanisms is essential for predicting microbial behavior in natural environments and biotechnological applications.
Table 1: Key Components of E. coli CCR and Inducer Exclusion
| Component | Function | Role in CCR/Inducer Exclusion |
|---|---|---|
| Enzyme IIAGlc | PTS protein for glucose transport | Central regulatory switch; dephosphorylated form inhibits non-PTS transporters [75] [73] |
| Adenylyl cyclase | Produces cAMP | Inhibited by unphosphorylated EIIAGlc; low cAMP reduces CAP activation [73] |
| CAP (Catabolite Activator Protein) | Transcription factor | Activated by cAMP; necessary for transcription of alternative sugar operons [73] |
| Lactose permease | Transports lactose into cell | Directly inhibited by unphosphorylated EIIAGlc [75] [76] |
| CcpA (in Gram+) | Catabolite control protein A | Mediates CCR in Gram-positive bacteria (e.g., Bacillus subtilis) [73] |
The integration of these regulatory constraints into metabolic models, particularly Flux Balance Analysis (FBA), presents significant challenges but is essential for accurately predicting bacterial growth and substrate utilization patterns [74] [27]. This protocol details methods for incorporating CCR and inducer exclusion mechanisms into genome-scale metabolic models of E. coli, enabling more realistic simulations of bacterial behavior in complex nutritional environments.
Biological Mechanisms of CCR and Inducer Exclusion
Molecular Basis of Catabolite Repression
The phosphotransferase system (PTS) serves as the central sensory apparatus for carbon quality in E. coli. When glucose is available, its transport through the PTS results in the dephosphorylation of EIIAGlc, creating the unphosphorylated form that initiates multiple inhibitory effects [73]. This dephosphorylation has two primary consequences: (1) inhibition of adenylate cyclase, reducing intracellular cAMP levels, and (2) direct binding to and inhibition of non-PTS sugar transporters (inducer exclusion) [75] [73].
The reduction in cAMP levels diminishes the formation of the cAMP-CAP complex, a global transcriptional activator required for the expression of numerous catabolic operons, including the lac operon [73]. This transcriptional regulation works in concert with pathway-specific repression mechanisms, such as the lac repressor protein, to ensure tight control of metabolic genes. Recent research has demonstrated that the combination of inducer exclusion and transcriptional regulation can lead to ~900-fold repression of lac expression when glucose is added to cells growing on glycerol and TMG [76].
Figure 1: CCR Regulatory Pathway in E. coli. Glucose transport through PTS triggers EIIAGlc dephosphorylation, inhibiting both adenylate cyclase and non-PTS transporters.
Expansion of Inducer Exclusion Mechanisms
While initially characterized for PTS sugars, inducer exclusion mechanisms extend beyond glucose. Research has demonstrated that glucose 6-phosphate, a non-PTS carbon source, can also cause inducer exclusion through similar mechanisms [75]. In these cases, glucose 6-phosphate causes dephosphorylation of EIIAGlc, leading to inhibition of lactose transport in wild-type strains but not in mutants lacking functional EIIAGlc or with EIIAGlc incapable of binding to the lactose carrier [75].
The potency of inducer exclusion is significantly enhanced in growing cells compared to cells treated with protein synthesis inhibitors like chloramphenicol. This amplification occurs through a positive feedback loop where declining intracellular inducer levels lead to reduced permease synthesis, which further reduces inducer uptake [76]. This system-level reinforcement makes inducer exclusion particularly effective in natural growing conditions.
Computational Framework for Integrating Regulatory Constraints
Foundation of Flux Balance Analysis
Flux Balance Analysis (FBA) is a constraint-based linear programming technique that predicts flow of metabolites through biochemical networks to optimize cellular objectives, typically biomass production [27]. The mathematical foundation of FBA involves:
- A stoichiometric matrix (S) representing all metabolic reactions
- A flux vector (v) containing reaction rates
- Constraint-based optimization: Maximize Z = cTv subject to Sv = 0 and vmin ≤ v ≤ vmax
The process of developing genome-scale metabolic models begins with genome annotation to identify metabolic genes, followed by reconstruction of metabolic networks connecting these genes to biochemical reactions [27]. For E. coli, several curated models exist with varying coverage of central carbon metabolism and regulatory systems.
Approaches for Integrating Regulatory Constraints
Two primary approaches have been developed for incorporating regulatory constraints into FBA frameworks:
Regulatory FBA (rFBA): Explicitly incorporates transcriptional regulatory rules that activate or inhibit reactions based on environmental and intracellular conditions.
Condition-Specific Constraints: Directly modifies flux bounds based on known regulatory effects, such as reducing uptake rates for non-preferred carbon sources during glucose availability.
For CCR and inducer exclusion, the following specific constraints can be implemented:
- Reduce maximum uptake rates for non-preferred carbon sources when preferred PTS sugars are present
- Constrain cAMP-dependent reactions based on extracellular glucose concentrations
- Implement Boolean regulatory rules that disable specific transporters when EIIAGlc is unphosphorylated
Table 2: Quantitative Parameters for CCR Modeling in E. coli
| Parameter | Value | Context | Reference |
|---|---|---|---|
| lac repression factor | ~900-fold | Growing cells on glycerol + TMG with glucose addition | [76] |
| Permease inhibition | 2-6 fold | Partial induction, chloramphenicol treated cells | [76] |
| Glucose uptake rate | 10.0 mmol/gDW/h | Typical maximal glucose uptake in E. coli | [77] |
| Lactose uptake (no glucose) | 6.5 mmol/gDW/h | Maximal lactose uptake without repression | [76] |
| Lactose uptake (with glucose) | 0.1-1.0 mmol/gDW/h | Lactose uptake during glucose repression | Estimated |
Protocol: Implementing CCR Constraints in FBA Models
Genome-Scale Model Reconstruction
Step 1: Genome Annotation and Reaction Identification
Begin with annotated genome data from RAST, PROKKA, or other annotation pipelines [27]. For E. coli PTS research, ensure specific annotation of:
- PTS system components (ptsI, ptsH, crr, ptsG)
- Sugar-specific EII complexes
- Non-PTS carbohydrate transporters
- Components of cAMP-CRP regulatory system
Step 2: Convert Functional Roles to Reactions
Map annotated genes to biochemical reactions using databases such as Model SEED. Pay special attention to:
- Enzyme complexes requiring multiple gene products
- Transport reactions for PTS and non-PTS carbohydrates
- Exchange reactions representing substrate uptake and secretion
Step 3: Define Metabolic Network and Biomass Objective
Compile all reactions into a stoichiometric matrix. Define appropriate biomass objective function reflecting E. coli macromolecular composition. Validate network connectivity, particularly around central carbon metabolism.
Incorporating CCR-Specific Constraints
Step 4: Implement PTS Transport Reactions
PTS reactions differ from standard transport as they simultaneously transport and phosphorylate substrates using phosphoenolpyruvate (PEP) as the phosphoryl donor:
Include appropriate ATP costs for PEP regeneration where needed.
Step 5: Add Regulatory Constraints for Inducer Exclusion
Modify flux bounds for non-PTS carbohydrate transporters based on glucose availability:
Step 6: Implement cAMP-CRP Regulatory Effects
Add constraints that reduce fluxes through CRP-dependent reactions when intracellular cAMP is predicted to be low. This can be implemented through:
- Boolean rules linking extracellular glucose to reaction activation
- Linear constraints that reduce maximum fluxes proportionally
Model Validation and Testing
Step 7: Simulate Diauxic Growth
Test the model in simulated media containing multiple carbon sources (e.g., glucose and lactose). The validated model should exhibit:
- Sequential substrate utilization with glucose consumed before lactose
- Characteristic diauxic growth curve with two distinct exponential phases separated by a lag period
- Appropriate metabolic fluxes during transition between phases
Step 8: Compare with Experimental Data
Validate model predictions against experimental growth data, substrate consumption rates, and gene expression patterns when available. Key validation datasets include:
- Growth curves in mixed substrate conditions
- Metabolite measurements during culture growth
- Fluxomics data from 13C labeling experiments
Figure 2: FBA Model Development Workflow. Key steps for building CCR-enabled metabolic models from genomic data.
Table 3: Key Research Reagent Solutions for CCR and PTS Studies
| Reagent/Resource | Function/Application | Example/Source |
|---|---|---|
| PyFBA | Python-based FBA modeling platform | Open-source package for building metabolic models from genome annotations [27] |
| Model SEED Biochemistry Database | Curated biochemical reactions | Comprehensive reaction database for model reconstruction [27] |
| RAST Annotation Server | Genome annotation | Identifies protein-encoding genes and assigns functional roles [27] |
| COBRA Toolbox | MATLAB-based metabolic modeling | Alternative FBA platform with regulatory constraint capabilities [27] |
| E. coli K-12 BW25113 | Model organism for PTS studies | Wild-type strain for CCR experiments [78] |
| E. coli pykA knockout | Mutant strain for model validation | Pyruvate kinase mutant for testing model predictions [77] |
| TMG (methyl-β-D-thiogalactoside) | Non-metabolizable lactose analog | Measures transporter activity without metabolism [75] [76] |
| Salicin | β-glucoside PTS substrate | Studies alternative PTS systems and CCR [79] |
Troubleshooting and Optimization
Common Implementation Issues
Problem: Unrealistic co-utilization of carbon sources
- Solution: Strengthen constraints on non-preferred substrate uptake. Implement more stringent flux bounds or add thermodynamic constraints.
Problem: Extended lag phases during substrate transitions
- Solution: Adjust the kinetics of regulatory switching mechanisms. Consider implementing dynamic FBA for more accurate transition modeling.
Problem: Underestimation of growth rates on secondary substrates
- Solution: Verify that cAMP-CRP activation is properly represented. Check that all essential enzymes for secondary substrate utilization are present in the model.
Model Refinement Techniques
- Parameter sensitivity analysis: Identify which parameters most significantly affect diauxic behavior
- Multi-strain validation: Test model predictions against multiple E. coli strains and mutants
- Integration with kinetic models: Incorporate kinetic parameters for key regulatory steps from established models [77] [78]
- Gap-filling procedures: Use automated algorithms to identify and correct missing metabolic functions [27]
Advanced Applications and Future Directions
Integrating CCR constraints into FBA models enables more accurate predictions of E. coli behavior in complex environments, with applications in:
- Metabolic engineering: Optimizing production of biofuels and biochemicals by manipulating carbon allocation
- Microbiome modeling: Predicting substrate preferences and competitive interactions between microbial species
- Bioprocess optimization: Designing feeding strategies that minimize metabolic conflicts and byproduct formation
Future developments should focus on incorporating multi-level regulatory information, including transcriptional, translational, and post-translational controls, to create more comprehensive models of bacterial decision-making [74] [78]. The integration of kinetic models of central carbon metabolism [77] with constraint-based approaches represents a promising direction for capturing both thermodynamic and enzymatic constraints.
The protocols outlined here provide a foundation for incorporating carbon catabolite repression and inducer exclusion into metabolic models, enabling more realistic simulations of E. coli growth and substrate utilization in complex environments.
Validating FBA Predictions and Comparing PTS Across Bacterial Species
Flux Balance Analysis (FBA) provides powerful predictions of metabolic behavior, but its outputs require experimental validation to ensure biological relevance. For research focused on the E. coli phosphotransferase system (PTS), benchmarking FBA predictions against 13C-Metabolic Flux Analysis (13C-MFA) data represents the gold standard for establishing predictive accuracy [80] [81]. This protocol details the integrated workflow for correlating these methodologies, enabling researchers to quantify the reliability of genome-scale model predictions and refine model parameters for enhanced fidelity in PTS studies.
Theoretical Foundation
Fundamentals of Flux Prediction and Estimation
Flux Balance Analysis (FBA) is a constraint-based modeling approach that predicts steady-state metabolic fluxes by optimizing a cellular objective (e.g., biomass yield) within the constraints of the metabolic network's stoichiometry [71] [82]. It provides predictions based on mathematical optimization, but these require validation. In contrast, 13C-Metabolic Flux Analysis (13C-MFA) estimates in vivo fluxes by integrating isotopic labeling data from 13C-tracer experiments with stoichiometric models [80] [83]. The fluxes obtained from 13C-MFA are considered a reliable reflection of the actual physiological state, making them an ideal benchmark for assessing FBA predictions [82] [84].
The Critical Need for Validation in PTS Research
The PTS in E. coli is a complex system involving carbohydrate uptake and regulatory interactions. FBA models of the PTS must accurately capture split ratios between glycolytic entry points and regulatory outcomes [85] [84]. However, FBA predictions can be influenced by:
- Incorrect objective function specification
- Incomplete network topology
- Inaccurate constraint definitions Systematic benchmarking against 13C-MFA directly addresses these limitations by providing experimental flux measurements for validation, particularly for central carbon metabolism where PTS operates [80] [82].
Experimental Design and Workflow
The following diagram illustrates the integrated workflow for benchmarking FBA predictions against 13C-MFA in E. coli PTS research:
Computational Methods for FBA
Model Selection and Preparation
For PTS-focused studies, begin with a well-curated genome-scale model of E. coli metabolism (e.g., iAF1260 or iJO1366) [84]. Essential preparation steps include:
- Verify complete representation of PTS reactions (Enzyme I, HPr, Enzyme II complexes)
- Confirm glucose uptake reaction through PTS is correctly modeled
- Check for alternative glucose transport systems (e.g., galP, mgt) that may operate under certain conditions
- Validate biomass composition equation relevance for your experimental conditions
Implementing FBA Simulations
Configure constraints to match planned experimental conditions:
- Carbon source: Glucose at physiologically relevant uptake rates
- Oxygen conditions: Aerobic or anaerobic as required
- Additional nutrients: Minimal media components
Advanced FBA Techniques for PTS Studies
For improved PTS flux predictions, consider these advanced FBA variants:
Regulatory FBA (rFBA): Incorporates Boolean rules for gene regulation affecting PTS components and carbon catabolite repression [85].
Integrated FBA (iFBA): Combines FBA with ordinary differential equations (ODEs) for dynamic processes; particularly useful for capturing PTS regulation and metabolite pooling effects [85].
Experimental 13C-MFA Protocol
Tracer Experiment Design for PTS Flux Resolution
For elucidating PTS-related fluxes in E. coli, employ specifically labeled glucose tracers:
Table 1: Tracer Selection for PTS-Related Flux Resolution
| Tracer Substrate | Label Position | Fluxes Resolved | Key PTS Insights |
|---|---|---|---|
| [1-13C] Glucose | C1 | Pentose phosphate pathway, Glycolysis | EIIA~P partitioning to regulation vs. transport |
| [U-13C] Glucose | Uniform | Complete central carbon metabolism | Comprehensive PTS flux into lower glycolysis |
| Parallel Labeling | Multiple tracers | Enhanced flux precision | PTS activity under different regulatory states |
Culture Conditions and Sampling
- Inoculum Preparation: Grow E. coli overnight in defined minimal medium with unlabeled glucose
- Experimental Culture: Dilute to OD600 ≈ 0.1 in fresh medium with 13C-labeled glucose
- Sampling for Metabolomics:
- Harvest 5-10 mL culture during mid-exponential growth (OD600 ≈ 0.5-0.8)
- Quick filtration or rapid centrifugation (<30 seconds) at 4°C
- Immediate quenching in cold methanol (-40°C)
- Store at -80°C until analysis
Analytical Methods for Labeling Measurements
Mass Spectrometry Analysis:
- Derivatize proteinogenic amino acids for GC-MS analysis
- Measure mass isotopomer distributions (MIDs) of key metabolites
- Focus on metabolites from glycolysis, PPP, and TCA cycle
- Technical replicates: n ≥ 3 for statistical reliability
Flux Calculation: Use specialized software (INCA, Omix) to estimate fluxes by fitting MIDs to metabolic network model [83] [82].
Data Integration and Benchmarking
Statistical Comparison Framework
Direct comparison requires normalization of flux values. Express fluxes as absolute rates (mmol/gDCW/h) or relative to glucose uptake rate. Key comparison metrics include:
Table 2: Key Fluxes for PTS Model Benchmarking
| Metabolic Reaction | FBA Prediction | 13C-MFA Estimate | Relative Difference | Physiological Significance |
|---|---|---|---|---|
| Glucose PTS uptake | Vpred | Vmfa | (Vpred - Vmfa)/Vmfa | Primary carbohydrate uptake |
| Phosphotransferase (EI/HPrt) | Vpred | Inferred | N/A | PTS protein phosphorylation |
| Pyruvate kinase flux | Vpred | Vmfa | (Vpred - Vmfa)/Vmfa | PEP/pyruvate balance |
| PPP flux (G6PDH) | Vpred | Vmfa | (Vpred - Vmfa)/Vmfa | NADPH production vs. PTS |
Correlation Analysis and Goodness-of-Fit
Calculate correlation coefficients (R²) between FBA-predicted and 13C-MFA measured fluxes. Use the χ²-test of goodness-of-fit to evaluate whether differences between predicted and observed labeling patterns are statistically significant [80]. A satisfactory correlation (typically R² > 0.85) indicates reliable FBA predictions for the PTS system under the tested conditions.
The Scientist's Toolkit
Table 3: Essential Research Reagents and Computational Tools
| Resource | Type/Category | Function in Benchmarking | Example Sources |
|---|---|---|---|
| [1-13C] Glucose | Tracer substrate | Enables flux estimation via 13C-MFA | Cambridge Isotopes |
| iAF1260, iJO1366 | Genome-scale model | Provides stoichiometric matrix for FBA | BiGG Models |
| INCA | Software platform | 13C-MFA flux estimation | (Young, 2014) |
| COBRA Toolbox | MATLAB package | FBA simulation and analysis | [83] |
| GC-MS System | Analytical instrument | Measures isotopic labeling | Various vendors |
| Escherichia coli K-12 | Model organism | PTS research model | ATCC |
Pathway Visualization of PTS and Central Metabolism
The following diagram illustrates the key metabolic pathways and fluxes in the E. coli PTS system and central carbon metabolism that are resolved through the benchmarking process:
Interpretation and Model Refinement
Analyzing Discrepancies
When FBA predictions significantly diverge from 13C-MFA estimates (relative difference > 20% for major fluxes), systematic investigation is required:
- PTS-Specific Issues: Check for incorrect representation of the phosphate transfer balance between PEP and pyruvate
- Regulatory Constraints: Verify if carbon catabolite repression is properly constrained in the model
- Energy Coupling: Examine ATP and redox (NADH/NADPH) balance around PTS reactions
- Alternative Routes: Identify if non-PTS glucose transport (e.g., via galP with subsequent glucokinase activity) is improperly constrained
Model Refinement Strategies
Based on benchmarking results, implement these refinement strategies:
Constraint Refinement:
- Adjust ATP maintenance requirements based on measured growth yields
- Modify PTS capacity constraints to match measured glucose uptake rates
- Implement regulatory constraints based on known PTS regulation
Objective Function Optimization: Test alternative objective functions if biomass maximization fails to predict accurate PTS fluxes:
- Minimization of metabolic adjustment (MOMA)
- Maximization of ATP yield
- Multi-objective optimization approaches
This protocol provides a comprehensive framework for benchmarking FBA predictions against 13C-MFA data in the context of E. coli PTS research. The rigorous correlation of computational predictions with experimental flux measurements establishes a cycle of model refinement and validation that enhances the predictive power of metabolic models. For PTS studies specifically, this approach enables researchers to quantify the accuracy of flux predictions through this critical uptake and regulatory system, ultimately supporting more reliable metabolic engineering strategies and deeper understanding of carbon catabolite regulation in E. coli.
The phosphoenolpyruvate-dependent phosphotransferase system (PTS) represents a remarkable multifunctional complex in bacteria that couples sugar transport with phosphorylation, serving as a central metabolic pathway and global regulatory network. This system employs phosphoenolpyruvate (PEP) as the phosphoryl donor to sequentially transfer phosphate groups through a series of proteins: enzyme I (EI), histidine-containing phosphocarrier protein (HPr), and substrate-specific enzyme II (EII) complexes [86]. The EII complexes typically consist of three components or domains: IIA, IIB, and the membrane-embedded IIC permease, with the mannose family incorporating an additional IID component [86]. Beyond its primary role in carbohydrate uptake and phosphorylation, the PTS participates in diverse cellular processes including carbon catabolite repression, chemotaxis, regulation of non-PTS transport systems, and virulence pathogenesis [86] [87].
Comparative genomic analyses have revealed striking diversity in PTS composition across bacterial species. Studies of 202 fully sequenced genomes identified PTS homologues exclusively within bacteria, with no evidence of horizontal transfer to archaea or eukaryotes [86] [88]. Among 174 bacterial genomes analyzed, approximately 44% possess the complete complement of PTS proteins required for sugar transport and phosphorylation, while 17% entirely lack PTS homologues, and another 17% retain cytoplasmic phosphoryl transfer proteins but lack recognizable permeases, suggesting dedicated regulatory functions [86]. The genomic allocation for PTS components can be substantial, with up to 3.2% of all genes in a bacterium encoding PTS proteins [86]. This application note details computational protocols for assessing PTS diversity and functional repertoire across bacterial genomes, providing a framework for integrating these analyses with flux balance analysis (FBA) models of bacterial metabolism.
Computational Workflow for PTS Analysis
The following workflow outlines a comprehensive approach for comparative analysis of PTS systems across bacterial genomes, from data acquisition through functional interpretation.
Diagram 1: Computational workflow for comparative analysis of bacterial PTS systems.
Genome Annotation and PTS Component Identification
The initial phase involves acquiring and annotating bacterial genomes to identify PTS components. High-quality genome sequences should be obtained from public databases such as RefSeq or PATRIC. Quality assessment is critical; genomes should have N50 ≥50,000 bp, CheckM completeness ≥95%, and contamination <5% [89]. Annotation can be performed using RAST for comprehensive subsystem-based annotation or Prokka for rapid annotation [90] [27]. RAST provides detailed functional assignments connected to the SEED database, facilitating direct mapping to metabolic subsystems including PTS components [27].
For PTS homology identification, employ sequence analysis tools such as BLAST or HMMER against custom databases of known PTS proteins. The PTS comprises several protein families, including enzyme I (EI), HPr, and various enzyme II complexes (IIA, IIB, IIC components) with distinct sugar specificities [86]. Iterative searches using PSI-BLAST or custom HMM profiles can enhance detection of divergent PTS homologues. It is essential to recognize that different strains of a single bacterial species often possess strikingly different complements of PTS proteins, highlighting the dynamic nature of PTS evolution [86] [88].
Classification and Functional Assignment
Identified PTS homologues should be classified into families based on substrate specificity and phylogenetic relationships. The major PTS permease families include glucose (Glc), fructose (Fru), lactose (Lac), galactitol (Gat), glucitol (Gut), mannose (Man), and ascorbate (Asc) families [86]. Structural analyses reveal that despite sequence diversity, PTS transporters share common architectural principles. For instance, the IIC domain typically forms a dimer with each monomer consisting of an N-terminal oligomerization domain and a C-terminal transport domain [15] [91].
Table 1: Major PTS Permease Families and Their Characteristics
| Family | Representative Substrates | Domain Organization | Structural Features |
|---|---|---|---|
| Glucose (Glc) | Glucose, Glucosides | IIA (separate), IICB (fused) | 8 transmembrane segments, cytoplasmic IIB domain [15] |
| Fructose (Fru) | Fructose, Mannitol | IIA (separate or fused), IICB (fused) | Varied domain fusions, elevator-type transport mechanism [91] |
| Lactose (Lac) | Lactose, N,N'-diacetylchitobiose | IIABC (fused or separate) | 10 transmembrane segments, reentrant helical hairpins [91] |
| Mannose (Man) | Mannose, Glucose | IIAB (fused), IIC (separate), IID (separate) | Additional IID component, unique specificity determinants [86] |
| β-Glucoside (Bgl) | β-Glucosides, Salicin | IIB, IIC, IIA (various arrangements) | Domain order affects function; BCA arrangement essential [91] |
Functional assignment of PTS components should extend beyond transport capabilities to include potential regulatory roles. Numerous PTS proteins participate in regulatory networks, controlling processes such as carbon metabolism, flagellar motility, and virulence gene expression [86] [87]. For example, in Salmonella, PTS components regulate virulence determinants associated with quorum sensing, pathogenicity islands, flagella, and the PhoPQ regulon [87]. The nitrogen-related phosphoryl transfer chain (EINtr, NPr, IIANtr) exemplifies PTS homologues that function exclusively in regulation without direct involvement in sugar transport [86].
Protocol: Integrating PTS Annotation with Metabolic Modeling
From Genomic Annotation to Reaction Network
The connection between genomic annotations and metabolic reactions forms the foundation for constructing genome-scale metabolic models. This process involves mapping identified PTS proteins to their corresponding biochemical reactions in the metabolic network.
Diagram 2: From genomic annotation to FBA simulation workflow.
Protocol Steps:
Convert Functional Roles to Reactions: Using platforms such as the Model SEED or PyFBA, map annotated PTS genes to their corresponding enzymatic reactions [27]. For example, the glucose-specific PTS transporter IICBGlc (encoded by ptsG) should be connected to the phosphotransferase reaction: PEP + glucoseₑₓₜₑᵣₙₐₗ → pyruvate + glucose-6-phosphateᵢₙₜₑᵣₙₐₗ [92] [27].
Account for Protein Complexes: Recognize that some PTS components form multi-protein complexes. For instance, enzyme I functions as a homodimer, while many EII complexes consist of multiple subunits [86] [27]. The PyFBA framework incorporates this through its complex-based reaction mapping, where multiple functional roles may be required for a single enzymatic activity [27].
Define Transport Reactions: For each identified PTS permease, include appropriate transport reactions in the model. PTS transport reactions differ from conventional transport as they involve vectorial phosphorylation without ATP consumption [15] [91].
Incorporate Regulatory Constraints: Integrate known PTS-mediated regulatory interactions into the model. For example, the phosphorylation state of IIAᴳˡᶜ influences non-PTS sugar uptake through allosteric regulation [86] [87].
Gap Filling and Model Validation
After constructing the initial model, perform gap filling to ensure metabolic functionality:
Identify Missing Functions: Compare the annotated PTS repertoire with expected metabolic capabilities. If a bacterium possesses PTS components for a specific sugar but lacks corresponding catabolic pathways, this represents a metabolic gap [27].
Add Essential Reactions: Using the Model SEED biochemistry database, identify and add reactions necessary for metabolic completeness [27]. For PTS models, pay particular attention to the connection between sugar phosphorylation and downstream metabolic pathways.
Validate with Experimental Data: Test model predictions against experimental observations. For example, if a bacterium grows on fructose but not glucose, the model should reflect this phenotype through its PTS composition and associated metabolic pathways [93] [92].
Research Reagent Solutions
Table 2: Essential Computational Tools for PTS Comparative Genomics
| Tool/Database | Function | Application in PTS Research |
|---|---|---|
| RAST | Genome annotation | Subsystem-based annotation of PTS components and related metabolic pathways [90] [27] |
| Model SEED | Metabolic model reconstruction | Connects PTS annotations to biochemical reactions; provides curated reaction database [27] |
| PyFBA | Flux balance analysis | Python library for building and analyzing metabolic models containing PTS reactions [27] |
| PATRIC | Comparative genomics | Pathway analysis and comparison of PTS systems across bacterial species [90] |
| HMMER | Protein family detection | Identification of divergent PTS homologues using custom profile hidden Markov models [89] |
| BRIG | Genome comparison | Visualization of PTS gene conservation across multiple bacterial genomes [90] |
| GapMind | Metabolic pathway analysis | Identification of missing metabolic steps in PTS-linked carbohydrate utilization pathways [27] |
Data Interpretation and Analysis
Comparative Genomic Patterns
Analysis of PTS distribution across bacterial genomes reveals several important evolutionary patterns. PTS protein phylogeny often differs from organismal phylogeny, indicating frequent horizontal gene transfer and lineage-specific loss of PTS components [86] [88]. Certain bacterial phyla, particularly Pseudomonadota, exhibit extensive PTS diversification, while others have minimal or specialized PTS complements [89]. The functional repertoire of PTS systems correlates with ecological niche specialization, with host-adapted pathogens often possessing distinct PTS profiles compared to environmental isolates [89].
Table 3: PTS Distribution Across Bacterial Ecological Niches
| Ecological Niche | PTS Characteristics | Functional Implications |
|---|---|---|
| Human-associated | Enriched for specific carbohydrate uptake systems | Adaptation to host carbohydrate availability; potential role in virulence [89] [87] |
| Animal-associated | Diverse PTS profiles; resistance gene associations | Zoonotic transmission potential; niche adaptation [89] |
| Environmental | Variable PTS complexity; metabolic versatility | Adaptation to diverse carbon sources in soil and aquatic environments [86] [89] |
| Clinical isolates | PTS linked to virulence regulation | Control of invasion, immune evasion, and persistence mechanisms [87] |
Structural and Functional Correlations
Structural studies of PTS components provide insights into functional mechanisms. Recent cryo-EM structures of the glucose transporter IICBGlc from E. coli reveal an elevator-type transport mechanism with substantial rigid body motion of the transport domain [15]. The IIC domain forms a dimer with each protomer consisting of eight transmembrane segments, and conformational changes alternate access to the substrate binding site between the periplasmic and cytoplasmic sides [15]. These structural insights inform our understanding of sequence-function relationships observed in comparative genomic analyses.
Conserved residues play critical roles in PTS function. For example, in the glucose superfamily of PTS transporters, a conserved histidine and glutamate appear to have shared roles in sugar binding and phosphorylation [91]. Understanding these conserved elements facilitates more accurate functional predictions from sequence data alone.
Troubleshooting and Optimization
Incomplete PTS Pathways: If genomic analysis reveals isolated PTS components without complete phosphoryl transfer chains, consider regulatory rather than transport functions. Many bacteria contain soluble PTS homologues that function exclusively in regulation [86] [88].
Discrepancies Between Annotators: Different annotation tools (RAST, PROKKA, BG7) may assign different functions to the same PTS component. Resolve conflicts by searching against curated databases and examining genomic context [90] [27].
Model Inconsistencies: When FBA predictions contradict experimental growth data, verify PTS reaction stoichiometry and ensure proper representation of phosphoryl transfer energetics [93] [92] [27].
Handling Domain Fusions: PTS components exhibit diverse domain arrangements across species. The EIIA, EIIB, and EIIC domains can exist as separate polypeptides or as fused proteins in various orders, which can affect function [91]. Account for these variations in functional predictions.
Application Notes
This document provides a detailed protocol for validating Flux Balance Analysis (FBA) predictions concerning the interaction of the Escherichia coli phosphotransferase system (PTS) with nitrogen and stress signaling pathways. The PTS is a major carbohydrate uptake system that also serves as a central regulatory network. While FBA can predict metabolic fluxes under different genetic and environmental conditions, these predictions require experimental validation to confirm the hypothesized cross-talk and regulatory interactions [31] [94]. These Application Notes outline a combined computational and experimental workflow to achieve this validation, focusing on the PTS(^{Ntr}) branch and its role in coordinating metabolic status with global physiology.
Table 1: Key FBA Predictions on PTS-Mediated Cross-Talk for Experimental Validation
| Predicted Interaction | Proposed Metabolic Link | Expected Phenotypic/Regulatory Outcome | Relevant FBA Constraints/Objectives |
|---|---|---|---|
| PTS(^{Ntr}) - Nitrogen Metabolism | PEP/pyruvate ratio reflecting carbon status influences nitrogen-related gene expression via PtsN phosphorylation [31]. | Altered growth yield on mixed carbon/nitrogen sources in PTS(^{Ntr}) mutants. | Incorporation of nitrogen assimilation costs (e.g., ATP for glutamine synthesis) into biomass objective function. |
| PTS - Potassium Homeostasis | PTS(^{Ntr}) controls potassium influx, indirectly affecting membrane potential and solute transport [31]. | Changes in tolerance to osmotic or ionic stress in ptsP, ptsO, or ptsN mutants. | Constraining potassium transport flux based on experimental uptake rates. |
| PTS - Global Stress Response | Integration of multiple metabolic inputs (PTS sugars, αKG, ATP) by the core PTS network [53]. | Defective transition to stationary phase or stress resistance in mutants. | Using a multi-objective optimization framework (e.g., maximizing growth and minimizing redox stress). |
Experimental Protocols
Protocol 1: Quantifying PTS Protein Phosphorylation States via Western Blot
1.1 Purpose: To experimentally measure the phosphorylation status of PtsN (EIIA(^{Ntr})) under different nutrient conditions, thereby validating FBA-predicted shifts in metabolic flux and the PEP/pyruvate ratio [31].
1.2 Materials:
- Wild-type E. coli and isogenic mutant strains (e.g., ΔptsN, ΔptsP).
- Minimal growth media with defined carbon sources (e.g., CAA, CAA+Fructose, CAA+Glucose) and nitrogen sources (e.g., Ammonia, Nitrate) [31].
- Lysis Buffer: 50 mM Tris-HCl (pH 7.5), 150 mM NaCl, 1% NP-40, supplemented with phosphatase inhibitors.
- Primary antibody specific for PtsN.
- HRP-conjugated secondary antibody.
- Chemiluminescent detection system.
1.3 Procedure:
- Culture Growth: Grow bacterial strains to mid-exponential phase in minimal media with the desired carbon/nitrogen sources.
- Sample Harvesting: Rapidly harvest cells by centrifugation (2 min, 10,000 x g, 4°C). Immediately freeze the pellet in liquid nitrogen to preserve phosphorylation states.
- Cell Lysis: Resuspend pellets in cold lysis buffer and disrupt cells by sonication. Clarify lysates by centrifugation.
- Western Blot: Separate proteins by SDS-PAGE and transfer to a PVDF membrane.
- Immunodetection: Probe the membrane with anti-PtsN antibody, followed by the secondary antibody. Develop using a chemiluminescent substrate. The phosphorylation state of PtsN is indicated by a mobility shift between its phosphorylated and non-phosphorylated forms [31].
- Data Analysis: Quantify band intensities to determine the ratio of phosphorylated to non-phosphorylated PtsN.
Protocol 2: Validating Metabolic Cross-Talk via FRET-Based Interaction Assays
2.1 Purpose: To monitor in vivo protein interactions within the PTS network in real-time, confirming its role as a global sensor that integrates sugar influx and other metabolic inputs [53].
2.2 Materials:
- E. coli strains expressing functionally tagged PTS proteins (e.g., EIIA(^{Glc})-CFP and EIICB(^{Mal})-YFP) from their native chromosomal loci [53].
- Microfluidic device or fluorescence-compatible plate reader.
- M9 minimal salts medium.
- Stock solutions of PTS sugars (e.g., glucose, mannitol) and non-PTS metabolites (e.g., α-ketoglutarate).
2.3 Procedure:
- Sample Preparation: Grow reporter strains to mid-exponential phase in M9 minimal medium.
- FRET Measurement: For plate reader assays, transfer cells to a 96-well plate. For microfluidic assays, load cells into the device. Continuously monitor CFP and YFP fluorescence.
- Stimulation: At a defined time point, inject a concentrated stock solution of a test compound (e.g., glucose, α-ketoglutarate) to achieve the desired final concentration.
- Data Acquisition: Record emission intensities at 475 nm (CFP) and 525 nm (YFP) every second for several minutes following stimulation.
- Data Analysis: Calculate the FRET ratio (YFP emission/CFP emission) over time. An increase in the FRET ratio indicates increased interaction between the two tagged proteins upon stimulation [53].
Diagram 1: Experimental workflow for validating FBA predictions.
The Scientist's Toolkit
Table 2: Essential Research Reagent Solutions
| Reagent / Tool | Function / Application | Key Characteristics & Examples |
|---|---|---|
| Genome-Scale Metabolic Models (GSMMs) | Platform for in silico FBA simulations to predict metabolic fluxes and generate testable hypotheses. | iJO1366 (E. coli): A comprehensive model containing PTS reactions. Requires manual curation to incorporate PTS(^{Ntr}) and regulatory constraints [95]. |
| FRET-Compatible Protein Tags | For monitoring dynamic protein-protein interactions within the PTS network in live cells. | CFP/YFP pairs: Used to tag PTS components (e.g., EIIA(^{Glc})-CFP, EIICB(^{Mal})-YFP) to measure stimulation-dependent recruitment [53]. |
| Phosphorylation-State Specific Tools | To directly assess the regulatory status of key PTS components like PtsN. | Anti-PtsN antibodies: Used in Western blots to detect mobility shifts corresponding to phosphorylated and non-phosphorylated forms [31]. |
| Defined Mutant Strains | To dissect the functional role of specific PTS branches and components in cross-talk. | PTS(^{Ntr}) mutants: ptsP, ptsO, ptsN knockout strains in P. putida and E. coli to study non-transport functions [31]. |
Integrated Data Analysis and Model Refinement
Table 3: Correlating Experimental Data with FBA Predictions
| Experimental Readout | FBA Prediction | Interpretation & Model Refinement Action |
|---|---|---|
| Decreased PtsN~P ratio upon fructose addition [31]. | High fructose uptake flux diverts phosphoryl groups from PTS(^{Ntr}) to PTS(^{Fru}). | Confirms model prediction of cross-talk. Action: Incorporate phosphate exchange reaction between the two PTS branches into the model. |
| Increased FRET between EIIA(^{Glc}) and EIICB(^{Mal}) upon glucose influx [53]. | The PTS network senses overall sugar influx, not just specific sugar identity. | Confirms integrated network function. Action: Constrain multiple sugar uptake rates simultaneously in FBA to reflect competition for shared phosphoryl groups. |
| Impaired growth yield of ΔptsN on poor nitrogen sources [31]. | PTS(^{Ntr}) links carbon status to nitrogen assimilation efficiency. | Validates regulatory role. Action: Integrate a regulatory rule (e.g., via RpoN) that modulates nitrogen uptake reactions based on PtsN phosphorylation state. |
Diagram 2: Signaling pathways of PTSNtr cross-talk.
The experimental data generated using these protocols should be used in an iterative feedback loop to refine the initial FBA model. For instance, quantitatively measured phosphorylation states of PtsN can be used to parameterize a kinetic module for the PTS, which can then be integrated with the stoichiometric GSM to create a more predictive hybrid model [94]. This iterative process of computational prediction and experimental validation is key to unraveling the complex, multi-level regulatory functions of the PTS.
Flux Balance Analysis (FBA) is a cornerstone of constraint-based metabolic modeling, using linear programming to predict steady-state metabolic flux distributions that optimize a cellular objective, such as biomass maximization [96] [27]. While FBA provides a powerful framework for simulating metabolism, its accuracy can be limited when predicting phenotypic responses across diverse genetic and environmental conditions [96] [97]. These limitations arise because traditional FBA often lacks mechanisms to incorporate regulatory effects and condition-specific constraints.
Hybrid modeling, which integrates machine learning (ML) with FBA, presents a paradigm shift. This approach enhances the predictive power of FBA models by using ML to derive context-specific constraints and parameters from multi-omic data, such as transcriptomics [96]. The resulting hybrid models achieve greater biological fidelity by capturing non-linear relationships and complex regulatory patterns that are difficult to represent in standard constraint-based models. For research on the Escherichia coli phosphotransferase system (PTS), a critical gateway for carbon uptake and metabolic regulation, this hybrid methodology offers a refined toolset to decipher the system's intricate influence on overall metabolic network function [98].
Computational Protocol: Regularized FBA Integrated with Machine Learning
This protocol provides a step-by-step guide for constructing a hybrid model to analyze E. coli PTS mutants. It integrates regularized FBA with machine learning to extract key features from transcriptomic and fluxomic data, enabling accurate prediction of metabolic adaptations.
Stage 1: Preparation of Metabolic Model and Multi-omic Data
- 1.1 Metabolic Model Curation: Begin with a high-quality, genome-scale metabolic model (GSMM) of E. coli. The model should be in a standard format (e.g., SBML). For PTS-focused studies, ensure the model accurately represents the PTS reactions for glucose transport and phosphorylation (encoded by ptsG, ptsHI, and crr), as well as alternative carbon uptake systems [98] [99].
- 1.2 Transcriptomic Data Processing: Acquire transcriptomic data (e.g., RNA-Seq) for both wild-type and PTS-mutant strains across multiple growth conditions. Process the raw read counts (e.g., RPKM/FPKM) to calculate fold-change values relative to a defined control condition (e.g., wild-type in minimal glucose medium). This normalization facilitates integration with flux data in subsequent analyses [96].
Stage 2: Condition-Specific Flux Estimation using Regularized FBA
- 2.1 Define Objective Functions and Constraints: For each strain and condition, set up the FBA simulation. Common objectives include maximization of biomass or ATP production. Incorporate the prepared transcriptomic fold-changes as constraints on the upper and lower bounds of reaction fluxes, typically using a method like E-Flux or a similar transformation [96].
- 2.2 Perform Regularized FBA: Execute a regularized FBA, such as parsimonious FBA, which finds a flux distribution that satisfies the objective while minimizing the total sum of absolute flux values. This approach helps eliminate metabolically irrelevant loops and produces a more biologically realistic flux distribution [96]. The output is a matrix of predicted reaction fluxes (v) for every reaction in the network across all experimental conditions.
Stage 3: Multi-omic Data Integration and Machine Learning Analysis
- 3.1 Create a Multi-omic Dataset: Concatenate the transcriptomic fold-change data and the predicted flux values for each reaction into a unified dataset, where each sample represents a condition and each feature is either a gene's transcript level or a reaction's flux [96].
- 3.2 Dimensionality Reduction and Clustering: Apply unsupervised learning techniques to the multi-omic dataset.
- Principal Component Analysis (PCA): Use PCA to reduce the dimensionality of the data and identify the principal components that explain the greatest variance across conditions. This helps visualize global metabolic responses and identify outliers [96].
- K-means Clustering: Perform k-means clustering on the principal component scores to group conditions with similar metabolic states. Use silhouette analysis to determine the optimal number of clusters [96].
- 3.3 Feature Selection using LASSO Regression: To identify the most important transcripts and fluxes that explain a key phenotypic output (e.g., growth rate), use LASSO (Least Absolute Shrinkage and Selection Operator) regression. LASSO performs variable selection by penalizing the absolute size of regression coefficients, driving less important features to zero [96]. The non-zero coefficients indicate transcripts or reactions that are strong predictors of the phenotype.
The following diagram illustrates the integrated computational workflow.
Experimental Validation Protocol for E. coli PTS Mutants
Computational predictions from hybrid models require experimental validation. This protocol outlines key wet-lab experiments to characterize an E. coli PTS- mutant (e.g., strain PB12, a ΔptsHIcrr mutant).
Physiological Characterization
- Objective: Quantify the growth phenotype and metabolic fluxes of the PTS- mutant compared to the wild-type strain.
- Method:
- Cultivation: Grow biological replicates of wild-type and PTS- strains in controlled bioreactors with defined M9 minimal medium supplemented with glucose as the sole carbon source. Maintain constant pH, temperature, and dissolved oxygen [98].
- Growth Kinetics: Monitor optical density (OD₆₀₀) throughout the fermentation to calculate growth rate (μ) and maximum biomass yield.
- Metabolite Analysis: Collect supernatant samples at regular intervals. Use High-Performance Liquid Chromatography (HPLC) or similar techniques to quantify the consumption of glucose and the production of metabolites (e.g., acetate, lactate, formate, ethanol) [98].
- Calculation of Specific Rates: Calculate specific glucose consumption rate (q₍Gₗc₎), specific acetate production rate (q₍Ace₎), and specific oxygen uptake rate (q₍O₂₎) based on the measured data.
Transcriptomic Validation via RT-qPCR
- Objective: Validate computational predictions of gene expression changes in central carbon metabolism.
- Method:
- Sample Collection: Harvest cells from the bioreactor during mid-exponential growth phase (e.g., OD₆₀₀ = 1.0) for RNA extraction [98].
- RNA Extraction and cDNA Synthesis: Extract total RNA and synthesize cDNA.
- RT-qPCR Assay: Perform Real-Time Quantitative PCR (RT-qPCR) for a panel of 96 genes involved in key pathways: glucose and acetate transport, glycolysis, gluconeogenesis, pentose phosphate pathway, TCA cycle, glyoxylate shunt, anaplerotic reactions, respiratory chain, and global regulators (e.g., cra, fis, arcA) [98].
- Data Analysis: Calculate relative gene expression using the 2^(-ΔΔCq) method, comparing the PTS- mutant to the wild-type strain.
Table 1: Key Physiological Parameters of E. coli PTS- Mutant (Representative Data)
| Strain | Growth Rate (h⁻¹) | qGlc (mmol/gDCW/h) | qAce (mmol/gDCW/h) | qO2 (mmol/gDCW/h) | ATP/ADP Ratio |
|---|---|---|---|---|---|
| Wild-type (JM101) | 0.50 | 8.5 | 2.1 | 12.0 | 8.2 |
| PTS- Mutant (PB12) | 0.38 | 10.2 | 3.5 | 15.5 | 6.5 |
Perturbing the Energy Charge
- Objective: Investigate the metabolic robustness of the PTS- mutant by perturbing its energy status.
- Method:
- Genetic Manipulation: Create a derivative strain (e.g., PB12AGD+) by overexpressing the atpAGD operon, which encodes the soluble F1 portion of the ATP synthase. This uncouples ATP hydrolysis in the cytoplasm, deliberately lowering the intracellular [ATP]/[ADP] ratio [98].
- Energetic Profiling: Measure the intracellular ATP and ADP concentrations in the mutant strain using a luminescence-based assay (e.g., ENLITEN ATP Assay) and calculate the [ATP]/[ADP] ratio [98].
- Systems Analysis: Correlate the perturbed energy state with the resulting physiological and transcriptomic profiles to understand how the PTS- mutant adapts to energetic stress.
The experimental workflow for generating validation data is summarized below.
The Scientist's Toolkit: Research Reagent Solutions
Table 2: Essential Reagents and Tools for Hybrid FBA-ML Studies in E. coli
| Item Name | Function/Description | Example/Reference |
|---|---|---|
| Genome-Scale Metabolic Model | A mathematical representation of E. coli metabolism for FBA simulations. | iML1515 (K-12 MG1655 model) [99]; iDK1463 (E. coli Nissle 1917 model) [99] |
| Automated Reconstruction Tool | Software for rapid generation of draft metabolic models from genomic data. | gapseq [100]; PyFBA [27]; CarveMe [100] |
| FBA Solver | Optimization software used to solve the linear programming problem in FBA. | GLPK, IBM CPLEX, COBRA Toolbox [27] |
| Machine Learning Library | Programming libraries for implementing PCA, clustering, and regression algorithms. | Scikit-learn (Python), Caret (R) [96] |
| PTS- Mutant Strain | Genetically defined E. coli strain lacking the phosphotransferase system components. | PB12 (ΔptsHIcrr derived from JM101) [98] |
| ATP/ADP Assay Kit | Bioluminescent kit for accurate quantification of intracellular adenine nucleotides. | ENLITEN ATP Assay System [98] |
Application Notes and Future Perspectives
The hybrid FBA-ML protocol reveals that PTS- mutants undergo significant metabolic rewiring. Key adaptations include upregulation of alternative glucose transporters (e.g., GalP), increased glycolytic and TCA cycle fluxes, and altered respiratory chain activity, which collectively compensate for the loss of the PTS [98]. The [ATP]/[ADP] ratio emerges as a critical regulator, influencing global gene expression and carbon flux distribution.
Future iterations of hybrid modeling can incorporate more advanced techniques. For instance, the TIObjFind framework uses topology-informed optimization to identify condition-specific metabolic objective functions, moving beyond static objectives like biomass maximization [97]. Furthermore, Dynamic FBA (DFBA) combined with methods like Partial Least Squares (PLS) regression can model the non-linear kinetics of metabolic reactions over time, providing a more dynamic view of the PTS- adaptation process [101]. These approaches, validated by the detailed experimental protocols herein, will continue to enhance our predictive understanding of microbial metabolism.
The phosphotransferase system (PTS) serves as a primary glucose uptake mechanism in many bacteria, including Escherichia coli, but its activity significantly influences intracellular phosphoenolpyruvate (PEP) pools and carbon flux distribution [24]. Flux Balance Analysis (FBA) has been extensively employed to model and understand the metabolic consequences of PTS manipulation in E. coli, revealing that PTS inactivation redirects carbon flux toward biosynthetic pathways, thereby enhancing precursor availability for aromatic amino acids and other valuable compounds [102] [103]. Corynebacterium glutamicum, an industrial workhorse for amino acid production, presents a unique case for applying these insights. While it possesses a PTS, its regulatory response to PTS disruption differs from E. coli, necessitating tailored engineering strategies [104] [105]. This protocol details the translation of PTS-focused FBA frameworks from E. coli to C. glutamicum, enabling the rational design of superior microbial cell factories for shikimate and derivative production.
Metabolic Background and Rationale
The Phosphotransferase System: A Dual-Function Mechanism
The PTS is a complex transport system that catalyzes the simultaneous uptake and phosphorylation of carbohydrates using phosphoenolpyruvate (PEP) as the phosphoryl donor [24]. The phosphoryl group from PEP is transferred through a conserved phosphorylation cascade: Enzyme I (EI) → Histidine-containing Phosphocarrier Protein (HPr) → Enzyme IIA (EIIA) → Enzyme IIB (EIIB) → the transported sugar bound to the membrane-bound Enzyme IIC (EIIC) [24]. In E. coli, PTS-mediated glucose import consumes one PEP molecule per glucose molecule internalized, creating direct competition between sugar uptake and biosynthesis of PEP-derived compounds. PTS inactivation in E. coli has been shown to increase PEP availability, which can be redirected toward the shikimate pathway for aromatic compound synthesis [103].
2C. glutamicum: Distinct Physiology and Engineering Potential
Unlike E. coli, C. glutamicum exhibits multiple glucose uptake systems, including a native non-PTS route comprising the myo-inositol transporter IolT1 and endogenous glucokinases (Glk1, Glk2, Ppgk) [104]. This provides a built-in redundancy that can be exploited when the PTS is inactivated. Furthermore, C. glutamicum demonstrates a high innate tolerance to aromatic compounds, making it an ideal platform for producing these often cytotoxic molecules [105]. The organism's lack of carbon catabolite repression (CCR) for many sugar mixtures further enhances its industrial applicability [105]. These distinctive physiological traits mean that while the fundamental FBA principles from E. coli PTS studies provide a valuable starting framework, their application to C. glutamicum requires specific modifications to account for its unique metabolic architecture.
Comparative Analysis of PTS Inactivation in E. coli and C. glutamicum
Table 1: Physiological and Metabolic Impacts of PTS Inactivation in E. coli vs. C. glutamicum
| Parameter | E. coli PTS- | C. glutamicum PTS- | Engineering Implications |
|---|---|---|---|
| Growth Rate | Dramatically reduced (μ = 0.1 h⁻¹ vs 0.71 h⁻¹ in wild-type) [103] | Significantly reduced [104] | Essential to implement growth restoration strategies for both organisms |
| Glucose Uptake Route | Galactose permease (GalP) + glucokinase (Glk) [103] | Native non-PTS system (IolT1 + glucokinases) or heterologous systems [104] | C. glutamicum possesses inherent redundancy; E. coli requires adaptation |
| Byproduct Formation | Acetate overflow minimized [102] | Not specifically reported | Reduced acetate in E. coli improves metabolic efficiency |
| PEP Availability | Increased [103] | Increased [104] | Benefits PEP-dependent products like shikimate in both hosts |
| Transcriptional Response | Upregulation of gluconeogenic genes (ppsA, pckA), TCA cycle genes [103] | Activation of non-PTS transporters and potential stress responses | Differential regulatory responses require host-specific tuning |
| Successful Production Examples | L-Phe (0.33 g/g glucose) [103] | Shikimate (4.1 g/L in flask, 141 g/L in fed-batch) [104] [105] | Both organisms demonstrate significant potential for aromatics |
Key Engineering Insights from Comparative Analysis
The comparative analysis reveals that while PTS inactivation in both organisms shares the common benefit of increased PEP availability, the implementation details differ substantially. E. coli PTS- strains typically require adaptive evolution to restore growth through enhanced non-PTS uptake systems [103], whereas C. glutamicum can leverage its innate non-PTS capacity more directly. The table also highlights the remarkable production potential of engineered C. glutamicum strains, with shikimate titers reaching 141 g/L in high-cell-density fermentation [105]. This demonstrates the significant payoff from properly implementing PTS-based engineering strategies in this organism.
Application Notes: C. glutamicum Engineering Framework
Integrated Metabolic Engineering Strategy
Based on FBA-guided principles from E. coli and validated in C. glutamicum, a successful engineering framework involves multiple coordinated interventions:
- PTS Inactivation: Delete ptsGHI operon to increase PEP availability [104].
- Growth Restoration: Implement one of two approaches:
- Pathway Optimization: Overexpress key shikimate pathway genes (aroGfbr, aroB, aroD, aroE) with feedback-resistant DAHP synthase [104] [105].
- Byproduct Elimination: Delete aroK (shikimate kinase) to prevent carbon diversion and create a shikimate-accumulating strain [105].
Table 2: Genetic Modifications for Enhanced Shikimate Production in C. glutamicum
| Target Module | Genetic Modification | Effect on Metabolic Network | Expected Outcome |
|---|---|---|---|
| Glucose Uptake | Delete ptsGHI | Increases intracellular PEP pool by eliminating PEP-consuming glucose import | Enhanced precursor supply for shikimate pathway |
| Delete iolR | Derepresses native non-PTS uptake (IolT1) | Improved glucose uptake and restored growth in PTS- strain | |
| Shikimate Pathway | Express feedback-resistant aroG (DAHP synthase) | Eliminates feedback inhibition by aromatic amino acids | Increased carbon flux into shikimate pathway |
| Overexpress aroB, aroD, aroE | Enhances conversion of DAHP to shikimate | Increased shikimate synthesis rate | |
| Carbon Diversion | Delete aroK (shikimate kinase) | Blocks conversion of shikimate to shikimate-3-phosphate | Shikimate accumulation; requires aromatic amino acid supplementation |
| Delete qsuB, qsuD | Prevents degradation of pathway intermediates | Improved carbon efficiency toward shikimate |
Quantifiable Outcomes of Multi-Modular Engineering
The combination of these strategies in C. glutamicum has yielded remarkable results. A study implementing PTS inactivation, iolR deletion, and pathway optimization achieved a shikimate content of 0.76 mg/mg DCW and a titer of 4.1 g/L in shake flask cultures [104]. More impressively, in high-cell-density fermentation, an engineered C. glutamicum R strain produced 141 g/L shikimate from glucose, demonstrating the tremendous industrial potential of this approach [105]. These results validate the FBA-informed strategy of manipulating PTS to optimize precursor availability for biosynthetic pathways.
Diagram 1: Metabolic Engineering Strategy for C. glutamicum Shikimate Production. This diagram illustrates the key genetic interventions (dashed lines) applied to optimize carbon flux from glucose to shikimate, including PTS deletion to increase PEP availability, IolR deletion to activate non-PTS glucose uptake, and shikimate pathway enhancement.
Experimental Protocols
Protocol 1: CRISPR/Cas9-Mediated Genome Editing in C. glutamicum
The development of advanced genetic tools has revolutionized metabolic engineering in C. glutamicum. This protocol describes efficient gene deletion using a two-plasmid CRISPR/Cas9 system [106] [107].
Materials:
- C. glutamicum ATCC 13032 or derivative strains
- pCas9 plasmid (constitutively expresses Cas9 nuclease)
- pgRNA plasmid (temperature-sensitive, expresses target-specific guide RNA)
- BHIS medium: Brain Heart Infusion with 0.5 M sorbitol
- NCM transformation buffer: 5 mM NaH₂PO₄, 5 mM K₂HPO₄, 10 mM 3-morpholinopropane-1-sulfonic acid (MOPS), 2 mM MgCl₂, 50 mM CaCl₂, 0.5 M sorbitol, pH 7.0
- Electroporation equipment
Procedure:
Design and Construction of Guide RNA:
- Identify target sequence (e.g., ptsG, ptsHI, iolR) with 5'-NGG PAM site immediately downstream
- Design 20-nucleotide spacer complementary to target region
- Synthesize and clone spacer into pgRNA plasmid using Golden Gate assembly
- Verify construct by sequencing [106]
Preparation of Competent Cells:
- Inoculate C. glutamicum in 5 mL BHIS medium, incubate overnight at 30°C with shaking
- Subculture 1:100 in fresh BHIS, grow to OD₆₀₀ ≈ 0.8-1.0
- Chill cells on ice for 30 minutes, harvest by centrifugation (5,000 × g, 10 min, 4°C)
- Wash twice with ice-cold NCM buffer
- Resuspend in minimal volume of NCM buffer (100-200 μL) [107]
Transformation:
- Mix 100-500 ng of each plasmid (pCas9 and pgRNA)
- Add to 100 μL competent cells, transfer to ice-cold 2-mm electroporation cuvette
- Electroporate (2.5 kV, 5 ms pulse for C. glutamicum ATCC 13032)
- Immediately add 1 mL BHIS medium, recover at 30°C for 2-4 hours
- Plate on selective media with kanamycin (pCas9) and chloramphenicol (pgRNA)
- Incubate at 30°C for 2-3 days [106] [107]
Mutant Verification:
- Screen colonies by colony PCR using primers flanking target region
- Verify successful deletion by DNA sequencing
- Cure plasmids by successive culturing at non-permissive temperature (37°C) without antibiotics [106]
Troubleshooting:
- Low transformation efficiency: Ensure proper cell washing and use freshly prepared electroporation buffers
- No mutant colonies: Verify guide RNA activity and include homologous repair template if necessary
- Plasmid curing difficulties: Perform multiple passages at elevated temperature with screening for antibiotic sensitivity
Protocol 2: Shikimate Production and Analysis
Materials:
- Engineered C. glutamicum shikimate production strain
- CGXII minimal medium: 40 g/L glucose, 20 g/L (NH₄)₂SO₄, 5 g/L urea, 1 g/L KH₂PO₄, 1 g/L K₂HPO₄, 0.25 g/L MgSO₄·7H₂O, 10 mg/L CaCl₂, 10 mg/L FeSO₄·7H₂O, 10 mg/L MnSO₄·H₂O, 1 mg/L ZnSO₄·7H₂O, 0.2 mg/L CuSO₄, 0.02 mg/L NiCl₂·6H₂O, 0.2 mg/L biotin, 30 mg/L protocatechuic acid [104]
- Aromatic amino acid supplement (as needed for auxotrophic strains): 100 mg/L L-phenylalanine, 100 mg/L L-tyrosine, 100 mg/L L-tryptophan
- HPLC system with refractive index or UV/Vis detector
Procedure:
Seed Culture Preparation:
- Inoculate single colony from fresh plate into 5 mL BHIS medium
- Incubate 16-24 hours at 30°C with shaking at 220 rpm
Production Culture:
- Inoculate seed culture into 25 mL CGXII medium in 250-mL baffled flask to initial OD₆₀₀ = 1.0
- Add required aromatic amino acids for auxotrophic strains
- Incubate at 30°C with shaking at 220 rpm for 48-72 hours [104]
Analytical Sampling:
- Withdraw 1 mL culture at regular intervals (e.g., 0, 12, 24, 48, 72 hours)
- Measure OD₆₀₀ for growth assessment
- Centrifuge at 13,000 × g for 5 minutes, collect supernatant for metabolite analysis
Shikimate Quantification by HPLC:
- Filter supernatant through 0.22-μm membrane
- Inject 10 μL onto SH1011 column (Shodex) or equivalent C18 reverse-phase column
- Use 10 mM H₂SO₄ as mobile phase at 0.6 mL/min, column temperature 75°C
- Detect shikimate by refractive index or UV at 210-220 nm
- Quantify using shikimic acid standard curve (0.1-10 g/L) [104]
Glucose and Byproduct Analysis:
- Analyze same samples for glucose consumption and organic acid byproducts (acetate, lactate, pyruvate)
- Use same HPLC conditions with appropriate standards [102]
The Scientist's Toolkit: Essential Research Reagents
Table 3: Key Reagents for C. glutamicum Metabolic Engineering
| Reagent/ Tool | Type | Function in Engineering | Examples/Notes |
|---|---|---|---|
| pK18mobsacB | Suicide vector | Allelic exchange via homologous recombination | Contains sacB for sucrose counter-selection [104] |
| CRISPR/Cas9 System | Genome editing | Targeted gene deletion/insertion | Two-plasmid system (pCas9 + pgRNA) [106] |
| pEC-XK99E | Temperature-sensitive vector | CRISPR sgRNA expression and editing template delivery | Contains temperature-sensitive repA [106] |
| RecT/RecET | Recombinase system | Enhances recombination efficiency with short homology arms | Enables efficient ssDNA recombineering [108] |
| Cpf1 (Cas12a) | CRISPR nuclease | Alternative to Cas9 with different PAM requirement | Used when Cas9 toxicity is problematic [108] |
| Non-PTS Transporters | Heterologous genes | Restores glucose uptake in PTS- strains | glf from Z. mobilis [104] |
| Feedback-resistant Enzymes | Pathway engineering | Deregulates metabolic flux | aroGfbr (DAHP synthase) [104] [105] |
Computational Modeling and FBA Integration
Diagram 2: FBA-Driven Metabolic Engineering Workflow. This iterative cycle begins with FBA model predictions from E. coli studies, informs C. glutamicum engineering strategies, validates predictions experimentally, and refines the models based on empirical data.
The successful translation of PTS engineering strategies from E. coli to C. glutamicum relies on a structured computational approach:
Model Construction and Constraining:
- Develop a genome-scale metabolic model of C. glutamicum incorporating PTS and non-PTS glucose uptake routes
- Apply constraints based on experimental data: glucose uptake rate, growth rate, byproduct secretion
- Implement PTS inactivation by setting the PTS flux to zero
Flux Prediction and Intervention Planning:
- Use FBA to predict flux redistribution following PTS deletion
- Identify potential bottlenecks in shikimate pathway and central metabolism
- Predict outcomes of combinatorial interventions (e.g., iolR deletion with pathway overexpression)
Model Refinement with Experimental Data:
- Incorporate ¹³C metabolic flux analysis (MFA) data to validate and refine model predictions
- Adjust kinetic parameters based on transcriptomic and metabolomic profiles
- Iteratively improve model accuracy through multiple engineering-design-test cycles [102]
This integrated computational-experimental approach dramatically accelerates the strain optimization process, reducing the traditional trial-and-error approach that has long hindered metabolic engineering efforts.
Conclusion
This FBA protocol establishes a robust framework for quantitatively analyzing the E. coli PTS, demonstrating its utility in predicting metabolic behavior, optimizing strains for biotechnology, and elucidating complex regulatory cross-talk. The integration of recent high-resolution structural data with computational models bridges a critical gap, providing mechanistic explanations for flux predictions. Future directions should focus on developing dynamic FBA (dFBA) models to capture transient metabolic states, incorporating structural insights into kinetic models, and expanding the framework to better predict how PTS manipulation influences the production of high-value compounds, such as shikimate and its derivatives. Ultimately, a refined understanding of the PTS through FBA will accelerate the design of next-generation bacterial cell factories and inform novel antimicrobial strategies targeting this unique bacterial system.
References
- 1. and homologies of carbohydrate: Structures ... phosphotransferase [https://pubmed.ncbi.nlm.nih.gov/1970652/]
- 2. - Wikipedia Phosphotransferase [https://en.wikipedia.org/wiki/Phosphotransferase]
- 3. PDB-101: Learn: Structural Biology Highlights: Bacterial... [https://pdb101.rcsb.org/learn/structural-biology-highlights/bacterial-phosphotransferase-system]
- 4. Analysis of global control of Escherichia coli carbohydrate ... [https://bmcsystbiol.biomedcentral.com/articles/10.1186/1752-0509-1-42]
- 5. Unbiased Quantitation of Escherichia coli Membrane ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2816013/]
- 6. What is flux balance analysis? - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC3108565/]
- 7. Cryo-EM structure of a phosphotransferase system glucose ... [https://www.sciencedirect.com/science/article/pii/S2590152425000054]
- 8. The Escherichia coli glucose transporter enzyme IICBGlc ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC133465/]
- 9. 9HNP: Cryo-EM structure of the glucose-specific PTS ... [https://www.rcsb.org/structure/9hnp]
- 10. 8QST: Cryo-EM structure of the glucose-specific PTS ... [https://www.rcsb.org/structure/8qst]
- 11. Electron crystallography reveals that substrate release from ... [https://www.sciencedirect.com/science/article/abs/pii/S1047847717300850]
- 12. A novel regulatory role of glucose transporter ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC314007/]
- 13. How phosphotransferase system-related protein ... [https://pubmed.ncbi.nlm.nih.gov/17158705/]
- 14. PEP group translocation [https://en.wikipedia.org/wiki/PEP_group_translocation]
- 15. Structure and mechanism of a phosphotransferase system ... [https://www.nature.com/articles/s41467-024-52100-3]
- 16. Metabolic Switching in the Sugar Phosphotransferase System ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1303199/]
- 17. Transcriptional and Metabolic Response of a Strain of ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11036233/]
- 18. Structure and function of the glucose PTS transporter from ... [https://www.sciencedirect.com/science/article/abs/pii/S1047847711002838]
- 19. Ten simple rules for creating reusable pathway models for ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8375987/]
- 20. Positive regulation of the pts operon of Escherichia coli [https://pmc.ncbi.nlm.nih.gov/articles/PMC207065/]
- 21. Modeling and analysis of flux distributions in the two branches ... [https://bmcsystbiol.biomedcentral.com/articles/10.1186/1752-0509-6-149]
- 22. The complete phosphotransferase system in Escherichia coli [https://pubmed.ncbi.nlm.nih.gov/11361063/]
- 23. KEGG ENZYME: 2.7.1.198 [https://www.genome.jp/dbget-bin/www_bget?ec:2.7.1.198]
- 24. How Phosphotransferase System-Related Protein ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1698508/]
- 25. Metabolic engineering of Escherichia coli for N-acetyl ... [https://pubmed.ncbi.nlm.nih.gov/41217679/]
- 26. From DNA to FBA: How to Build Your Own Genome-Scale ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4911401/]
- 27. From DNA to FBA: How to Build Your Own Genome-Scale ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2016.00907/full]
- 28. Genome-Scale Metabolic Model of Xanthomonas phaseoli pv ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7432306/]
- 29. Reconstruction and application of a genome-scale metabolic ... [https://bmcgenomics.biomedcentral.com/articles/10.1186/s12864-025-12195-4]
- 30. The Biomass Objective Function - PMC - PubMed Central [https://pmc.ncbi.nlm.nih.gov/articles/PMC2912156/]
- 31. Modeling and analysis of flux distributions in the two branches ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC3562155/]
- 32. Tryptophan Production Maximization in a Fed-Batch Bioreactor with... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8698263/]
- 33. sciencedirect.com/science/article/pii/S0362028X22067102 [https://www.sciencedirect.com/science/article/pii/S0362028X22067102]
- 34. Cryo-EM structure of a phosphotransferase system glucose ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11930441/]
- 35. A genome-scale Escherichia coli kinetic metabolic model k ... [https://www.nature.com/articles/ncomms13806]
- 36. BiGG Reaction SUCpts in iJR904 [http://bigg.ucsd.edu/models/iJR904/reactions/SUCpts]
- 37. Universal reaction: SBTpts [http://bigg.ucsd.edu/universal/reactions/SBTpts]
- 38. Quantifying the propagation of parametric uncertainty on ... [https://www.sciencedirect.com/science/article/pii/S1096717621001634]
- 39. Universal reaction: SUCpts [http://bigg.ucsd.edu/universal/reactions/SUCpts]
- 40. Metabolic Modeling of Common Escherichia coli Strains in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4122010/]
- 41. Glucose Transport in Escherichia coli - PubMed Central - NIH [https://pmc.ncbi.nlm.nih.gov/articles/PMC10305011/]
- 42. Phosphoenolpyruvate:carbohydrate phosphotransferase ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC372926/]
- 43. Elementary Mode Analysis: A Useful Metabolic Pathway ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2909134/]
- 44. Hierarchy of non-glucose sugars in Escherichia coli [https://bmcsystbiol.biomedcentral.com/articles/10.1186/s12918-014-0133-z]
- 45. Regulation of gene expression in the PTS in Escherichia coli [https://www.sciencedirect.com/science/article/abs/pii/S1369527402002965]
- 46. Inducer exclusion in Escherichia coli by non-PTS substrates [https://pubmed.ncbi.nlm.nih.gov/9822815/]
- 47. Growth strategy of microbes on mixed carbon sources - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC6427025/]
- 48. Glucose repression can be alleviated by reducing ... [https://www.nature.com/articles/s41598-018-20804-4]
- 49. Physiological Regulation of the Derepressible Phosphate ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC107158/]
- 50. Identification and functional characterization of a fructose ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC11837500/]
- 51. Predicting microbial interactions with approaches based on ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/s12859-024-05651-7]
- 52. Current knowledge of the Escherichia coli ... [https://pubmed.ncbi.nlm.nih.gov/22573269/]
- 53. Sugar Influx Sensing by the Phosphotransferase System of ... [https://journals.plos.org/plosbiology/article?id=10.1371/journal.pbio.2000074]
- 54. The protein–protein interaction network of the Escherichia ... [https://www.sciencedirect.com/science/article/pii/S0923250821000966]
- 55. EColiCore2: a reference network model of the central ... [https://www.nature.com/articles/srep39647]
- 56. Robustness Analysis of the Escherichia Coli Metabolic ... [https://pubmed.ncbi.nlm.nih.gov/11101318/]
- 57. Core E. coli Model [https://systemsbiology.ucsd.edu/Downloads/E_coli_Core]
- 58. The Bacterial Phosphotransferase System: New frontiers ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC4512285/]
- 59. Modeling and experimental validation of the signal ... [https://www.sciencedirect.com/science/article/abs/pii/S0168165604000914]
- 60. Model-Based Characterization of E. coli Strains with Impaired ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10376147/]
- 61. A Quantitative Approach to Catabolite Repression in ... [https://www.sciencedirect.com/science/article/pii/S0021925820706782]
- 62. Experimental determination of Escherichia coli biomass ... [https://journals.plos.org/plosone/article?id=10.1371/journal.pone.0262450]
- 63. Chapter 25 Phosphotransferase systems or PTSs as ... [https://www.sciencedirect.com/science/article/abs/pii/S1383812196800662]
- 64. Sugar transport. Properties of mutant bacteria defective in ... [https://pubmed.ncbi.nlm.nih.gov/789368/]
- 65. Sugar transport. Properties of mutant bacteria defective in ... [https://www.sciencedirect.com/science/article/pii/S0021925817329873]
- 66. Evolution of gene knockout strains of E. coli reveal ... [https://www.nature.com/articles/s41467-018-06219-9]
- 67. pts promoter influences antibiotic resistance via proton ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2023.1276954/full]
- 68. Characterization of physiological responses to 22 gene ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5845446/]
- 69. A novel knock out strategy to enhance recombinant protein ... [https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-020-01407-z]
- 70. Enzyme I facilitates reverse flux from pyruvate to ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC5290146/]
- 71. Metabolic flux balance analysis and the in silico ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC29061/]
- 72. Flux balance analysis [https://en.wikipedia.org/wiki/Flux_balance_analysis]
- 73. Catabolite repression [https://en.wikipedia.org/wiki/Catabolite_repression]
- 74. Understanding carbon catabolite repression in Escherichia ... [https://pubmed.ncbi.nlm.nih.gov/25475882/]
- 75. Inducer exclusion by glucose 6-phosphate in Escherichia coli [https://pubmed.ncbi.nlm.nih.gov/9643543/]
- 76. Inducer exclusion, by itself, cannot account for the glucose- ... [https://www.sciencedirect.com/science/article/pii/S0006349522000455]
- 77. Kinetic modelling of central carbon metabolism in ... [https://pubmed.ncbi.nlm.nih.gov/22823407/]
- 78. Modeling the interaction between the central carbon ... [https://www.sciencedirect.com/science/article/abs/pii/S1369703X20303077]
- 79. Phosphotransferase System Uptake and Metabolism of the ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC7504967/]
- 80. Model Validation and Selection in Metabolic Flux Analysis and ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10922127/]
- 81. Metabolic flux analysis and visualization [https://pubmed.ncbi.nlm.nih.gov/21815690/]
- 82. A Method to Constrain Genome-Scale Models with 13 C ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1004363]
- 83. Systematic comparison of local approaches for isotopically ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10280729/]
- 84. 13C metabolic flux analysis at a genome-scale [https://www.sciencedirect.com/science/article/abs/pii/S1096717615001032]
- 85. Integrating metabolic, transcriptional regulatory and signal ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC6702764/]
- 86. Comparative Genomic Analyses of the Bacterial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC1306802/]
- 87. Phosphotransferase system in regulation of Salmonella ... [https://www.sciencedirect.com/science/article/pii/S0944501318310668]
- 88. Comparative genomic analyses of the bacterial ... - PubMed [https://pubmed.ncbi.nlm.nih.gov/16339738/]
- 89. Comparative genomics reveals key adaptive mechanisms ... [https://www.frontiersin.org/journals/microbiology/articles/10.3389/fmicb.2025.1543610/full]
- 90. Tools for bacterial comparative genomics [https://holtlab.net/2015/02/25/tools-for-bacterial-comparative-genomics/]
- 91. Structural insight into the PTS sugar transporter EIIC - PMC [https://pmc.ncbi.nlm.nih.gov/articles/PMC4169766/]
- 92. Modeling and simulation of the main metabolism in ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC2999585/]
- 93. Metabolic flux balance analysis and the in silico analysis of ... [https://bmcbioinformatics.biomedcentral.com/articles/10.1186/1471-2105-1-1]
- 94. Current status and future perspectives of kinetic modeling for ... [https://bioresourcesbioprocessing.springeropen.com/articles/10.1186/s40643-014-0031-7]
- 95. Flux balance analysis-based metabolic modeling of microbial ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10449171/]
- 96. Protocol for hybrid flux balance, statistical, and machine ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC8488602/]
- 97. Optimization-based framework with flux balance analysis (FBA ... [https://journals.plos.org/ploscompbiol/article?id=10.1371/journal.pcbi.1013635]
- 98. Transcriptional and Metabolic Response of a Strain ... [https://www.mdpi.com/2673-6284/13/2/10]
- 99. Development of a Genome-Scale Metabolic Model and ... [https://www.mdpi.com/1422-0067/22/4/2122]
- 100. gapseq: informed prediction of bacterial metabolic pathways ... [https://genomebiology.biomedcentral.com/articles/10.1186/s13059-021-02295-1]
- 101. Hybrid dynamic flux balance modeling approach for ... [https://pubmed.ncbi.nlm.nih.gov/40133554/]
- 102. Metabolic perturbations in mutants of glucose transporters and ... [https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-019-1224-8]
- 103. Metabolic transcription analysis of engineered Escherichia ... [https://microbialcellfactories.biomedcentral.com/articles/10.1186/1475-2859-6-30]
- 104. The effect on shikimate production by deleting iolR and ... [https://www.frontiersin.org/journals/bioengineering-and-biotechnology/articles/10.3389/fbioe.2025.1616558/full]
- 105. Engineering of Corynebacterium glutamicum for the synthesis ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC12125101/]
- 106. Efficient gene editing in Corynebacterium glutamicum using ... [https://microbialcellfactories.biomedcentral.com/articles/10.1186/s12934-017-0814-6]
- 107. CRISPR/Cas9-mediated ssDNA Recombineering in ... [https://en.bio-protocol.org/en/bpdetail?id=3038&type=0]
- 108. Synthetic biology tools for engineering Corynebacterium ... [https://pmc.ncbi.nlm.nih.gov/articles/PMC10024154/]